Junction Dysfunction Basics
I’d argue a lot of common health issues are due to something I’ve coined as “Junction Dysfunction” (JD).
JD consists of Transient Capillary Leak Syndrome (TCLS) and Micro-Sepsis (MSS) and in this post we will be discussing the former: TCLS.
CLS and TCLS
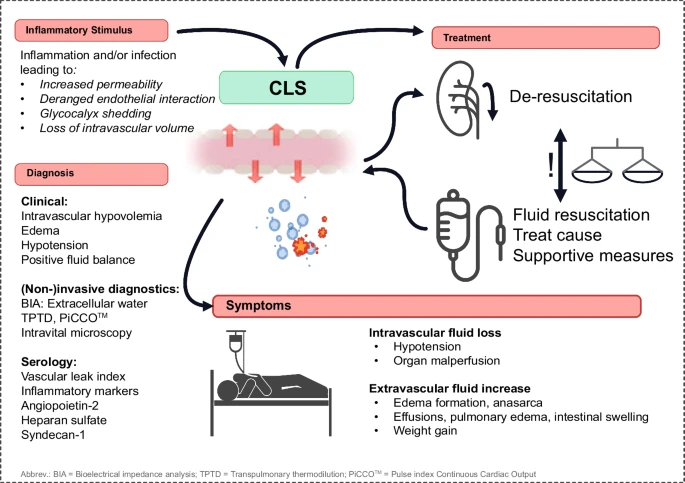
Capillary leak syndrome (CLS) in clinical practice, is frequently defined by excessive fluid shift from the intravascular to the extravascular space, resulting in intravascular hypovolemia, extravascular edema formation, and hypoperfusion. R
CLS can be triggered by numerous disease states as well as certain medications and toxins. R
Depending on the literature source, terms like “generalized hyperpermeability syndrome”, “endothelial permeability” or “capillary leakage” may be used synonymously for CLS. R
When CLS happens on a micro/transient level, this is what I call Transient Capillary Leak Syndrome (TCLS).
What Triggers TCLS?
This is not an exclusive list: R
Cardiopulmonary bypass
Anaphylaxis
Major burns
Ovarian hyper stimulation syndrome
Hemophagocytic lymphohistiocytosis
Viral hemorrhagic fevers
Autoimmune diseases
Snakebite envenomation
Ricin poisoning
IL's
Monoclonal antibodies
Gemcitabine
Anti-thymocute glocilon
The Endothelium, Leakage and Capillaries
Endothelium
The endothelium is a single layer of cells lining the interior surface of all blood vessels. R
Its surface area has been estimated to be equivalent to a soccer field! R
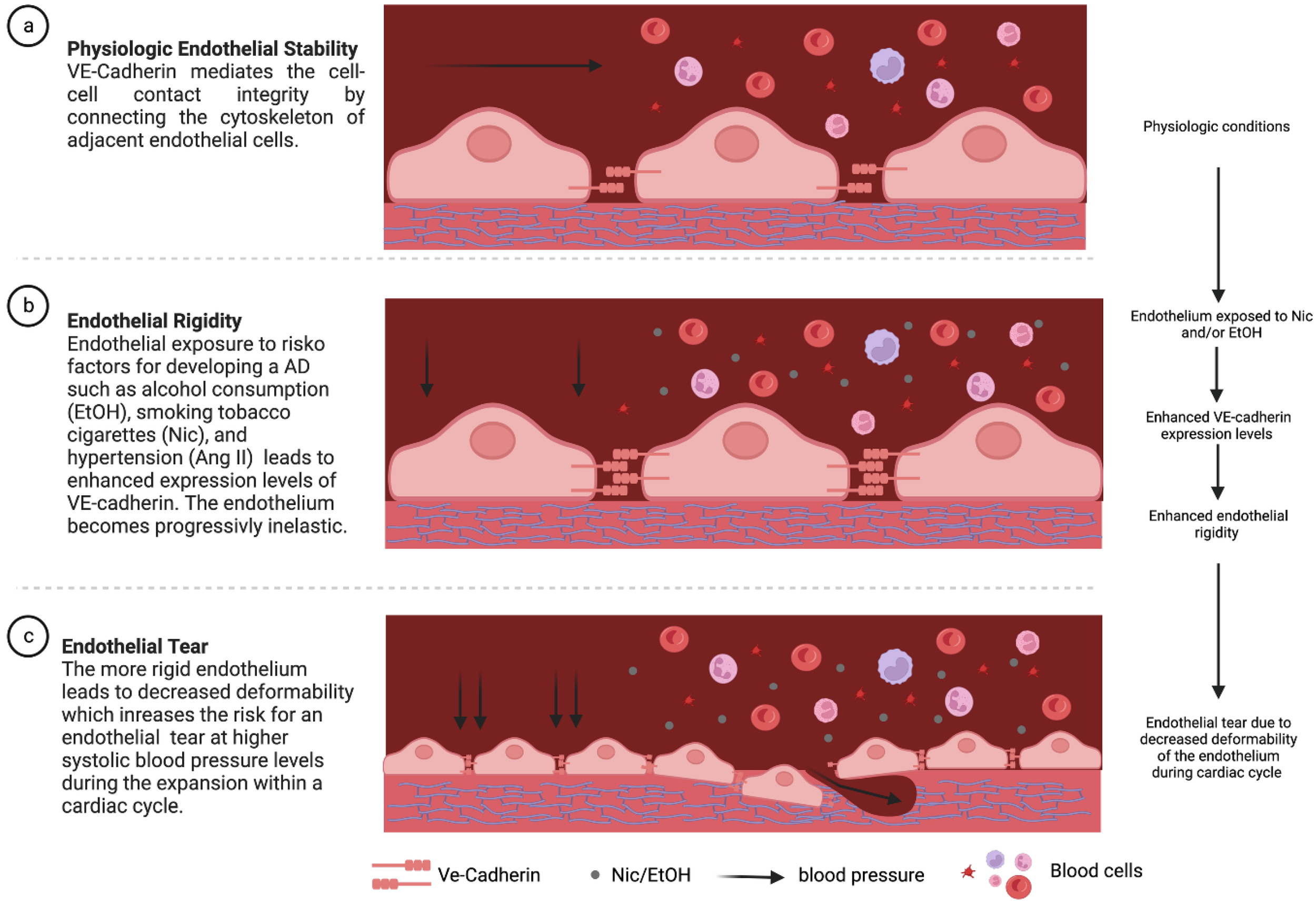
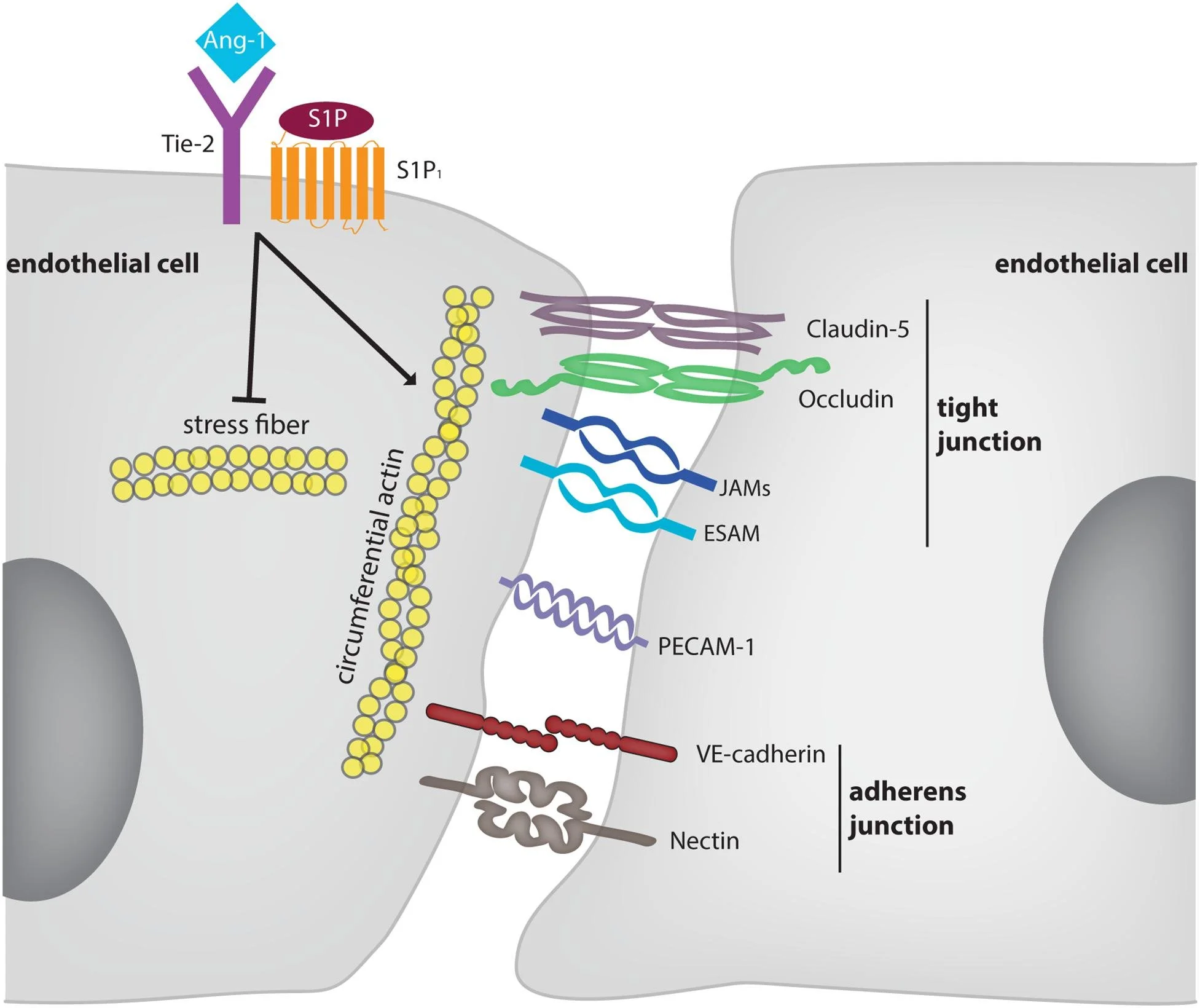
Anyway, the way the endothelial cells of endothelium are able to regulate permeability - largely due to their ability to form tight, adherens, and gap junctions, the latter of which allow for the exchange of ions, various metabolites, and regulatory factors. R
The endothelium acts as a semi-permeable barrier, controlling the exchange of macromolecules and fluids between interstitial fluid and blood. R
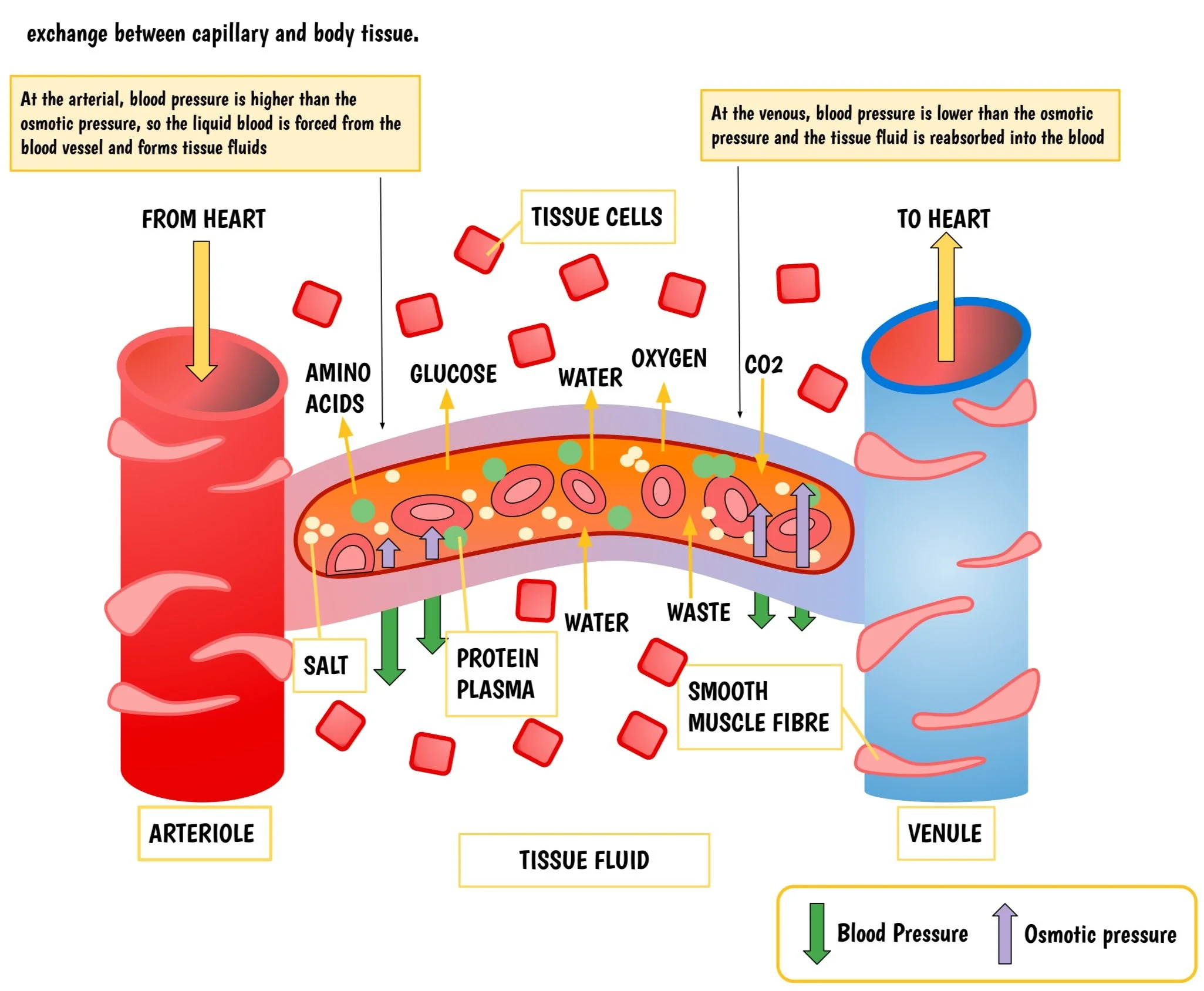
Vascular Leakage Pathways
wikipedia - @Julia222t
The function of the endothelial barrier varies across different segments of the microvasculature, with permeability increasing from arterioles to venules. R
For example, the venules possess great permeable abilities (allowing for leukocytes to exit the bloodstream), while large veins are less prone to fluid leakage (don’t respond to agents that increase permeability). R
When there is TCLS, there is vascular leakage. R
Vascular leakage can occur through two primary pathways: R
Paracellular
Transcellular
Capillary Subtypes
Continuous
Fenestrated - diameter of 60 nm, their permeability is primarily restricted to water and minor hydrophilic solutes
Discontinuous
Inflammation, Infections, Glycocalyx, and TCLS
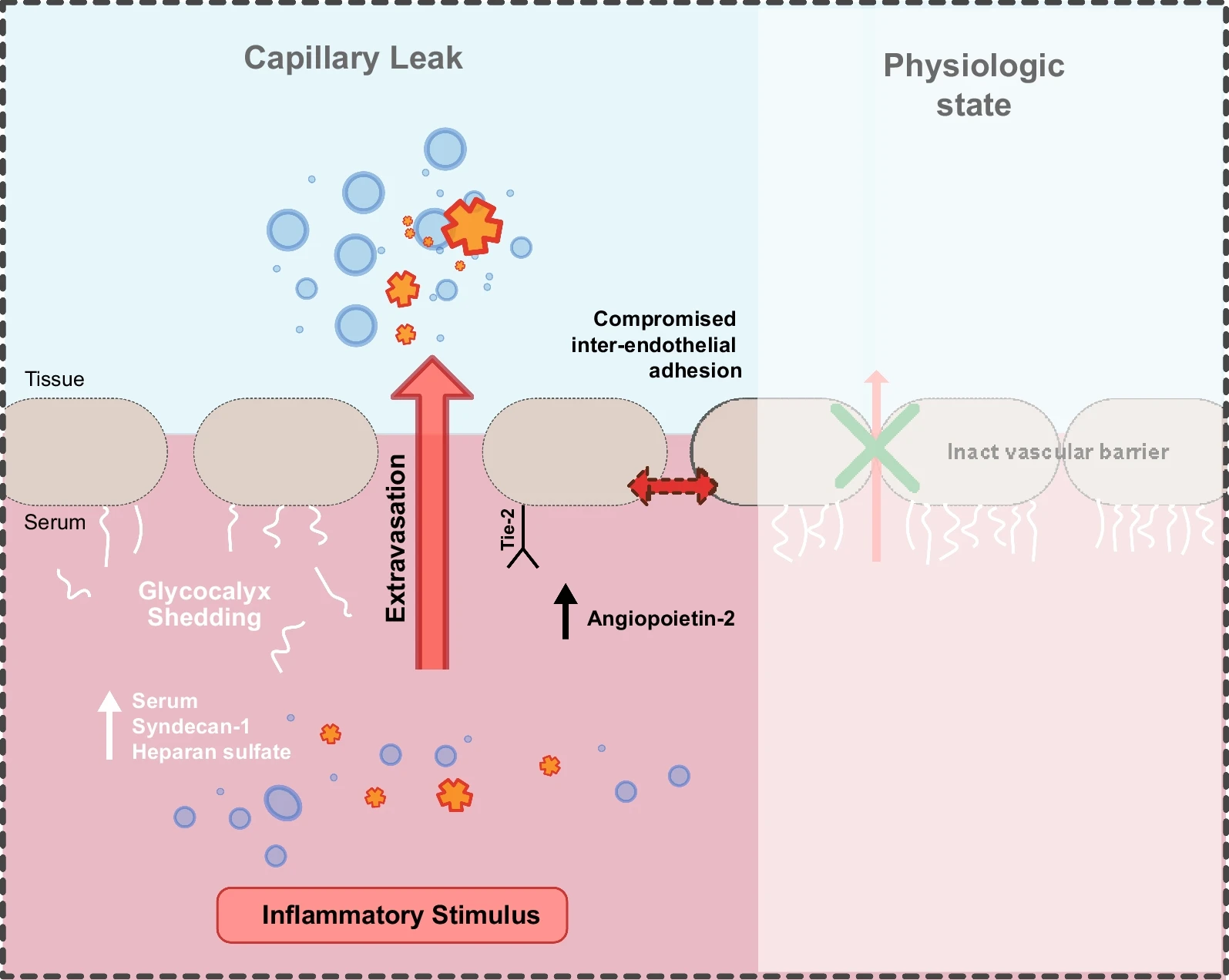
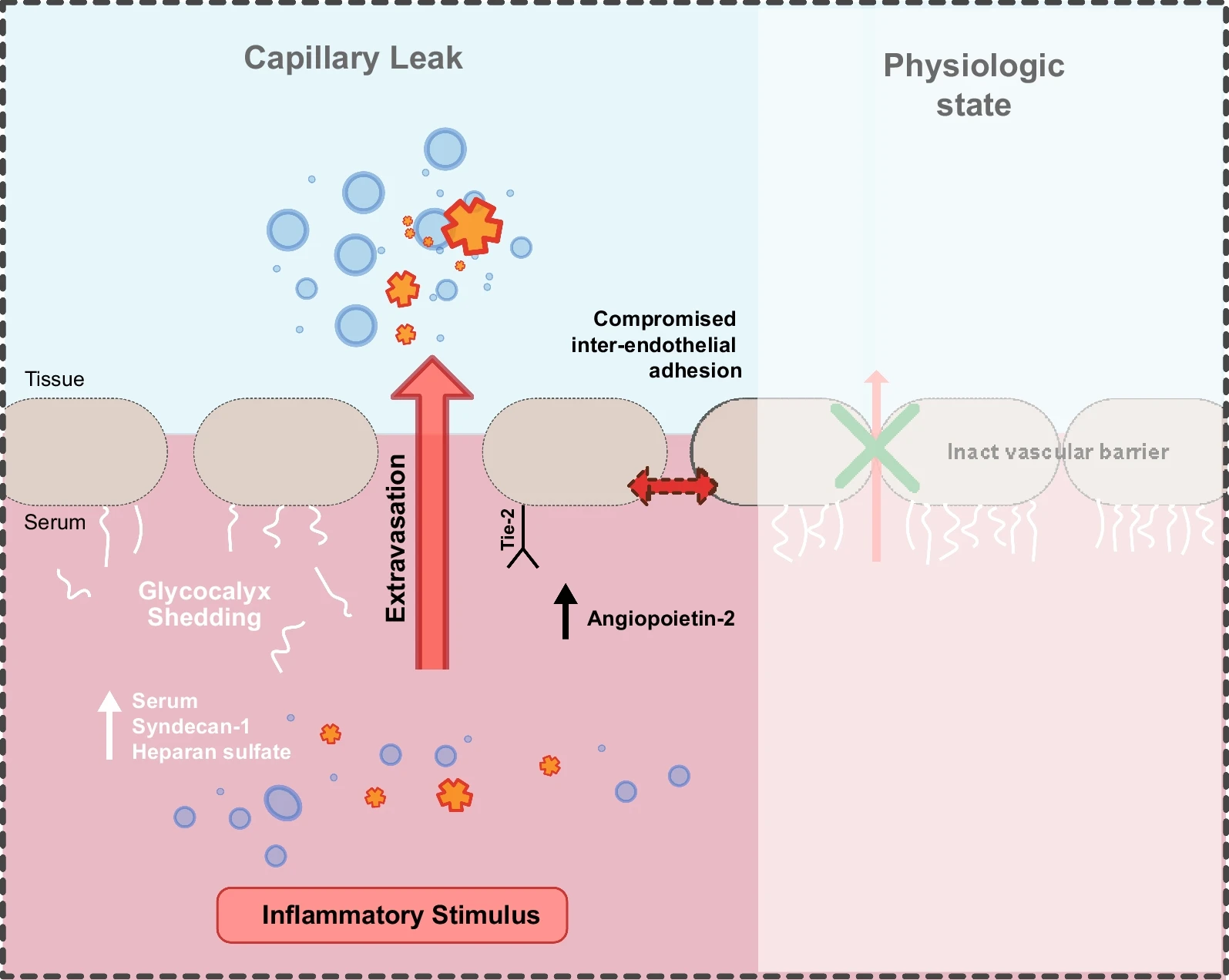
When something inflammatory happens (like stimulation of the immune system from PAMPs or DAMPs), immune cells use shedding (HA-ase and MMPs) to breakdown the glycocalyx. R R
This breaks open your tight junctions and allows for fluid to leak into the tissue. R
Not only that, endothelial cells release proteins such as tissue factor, P-selectin, von Willebrand factor, interleukins, angiopoietin-2 (Ang-2) and many more into the bloodstream. R
The aim for releasing all of these into the blood stream is to attract immune cells to the localized sites of infection of damage. R
This is okay locally, but when this proinflammatory response escalates to a systemic level, it can lead to a widespread compromise of the endothelial barrier function and cause massive Junction Dysfunction. R
This results in TCLS with relevant fluid shifts, hypotension, intravascular hypovolemia, edema formation. R
Types Of Adherin Tight Junctions
This is not an exhaustive list. R
| Interactors |
Function |
|---|---|
| β-catenin | junction architecture and signaling |
| p120 | VE-cadherin stabilization and signaling |
| plakoglobin | junction architecture and signaling |
| α-catenin, α-actinin, eplin, N-WASP | cytoskeletal anchorage and organization |
| Junction Stability and Cytoskeleton Organization | |
| Tiam | activation of Rac, junction stability |
| Rap1, Raf1, MAGI | junction stability and maturation |
| Phosphatases Junction Stability | |
| VE-PTP | VE-cadherin dephosphorylation, permeability control, leukocyte diapedesis |
| PTP1B, PTP2A, SHP2, DEP-1, RPTP-μ | permeability control |
| Csk | Src inactivation, inhibition of cell growth |
| Cell Polarity | |
| PAR3/PAR6/aPKC | cell polarity, lumen formation |
| CCM1/Krit1 | cell polarity, junction stability |
| Growth Factor Receptors | |
| VEGFR2 | contact inhibition of cell growth, permeability control |
| FGF-R | cell motility and growth |
| TGFβ-R | inhibition of cell motility |
| Induction of Permeability | |
| β-arrestin1, β-arrestin2, Caveolin1 | junction permeability |
| Other Functions | |
| PECAM | mechanosensor |
| PI3 kinase, Akt, Shc | signal transduction |
| Src, FAK | increase in permeability |
Angpt Tie2 axis and tight junctions (ve-cadherin)
Angiopoietins (ANGPT - ANG1 and ANG2) are essential for regulating vascular integrity.
They work in conjunction with receptor tyrosine kinases (RTKs) Tie1 (TIE) and Tie2 (TEK).
Through their regulator mechanisms they help facilitate: R R R
Blood vessel stabilization and remodeling
Modulation of vascular permeability
Blood and lymphatic vascular homeostasis
Angiogenesis
Lymphangiogenesis
Inflammation
What this means is that they help or hurt endothelial cells and promote the maturation and stability of blood vessels. R
Tie2 signaling is highly active when blood vessels are healthy - providing stable junctions, anti-inflammatory effects, and anti-coagulant properties. R
ANGPT/TIE2 In Hypoxia
When the glycocalyx becomes degraded or thins out, ANGPT/TIE2 gets destabilized, due to hypoxia. R
Not only that but in reverse, when ANGPT/TIE2 gets destabilized, this leads to very fast shedding of the glycocalyx (via heparanase (HA-ase) release). R
During this dysregulation, especially during an infection, there is a loss of ANGPT1, with an increase of ANGPT2. R R
For example, with Long Covid and ME/CFS, there is higher levels of ANGPT2, consistent with glycocalyx loss. R
The good news is that by doing all of these remodeling techniques (like this whole series is about), we can restore the glycocalyx back to favorable dimensions. R
So next we need to discuss Hypoxia Inducible Factors (HIF1a and HIF2a, specifically).
HIF1a and HIF2a
Hypoxia Inducible Factor 1 alpha and Hypoxia Inducible Factor 2 alpha are always active and they come from out mitochondria. R
In normal aerobic respiration, they are quickly degraded by enzymes, but when you’re in a hypoxic state, those enzymes are no longer able to break down HIFs. R
Maybe you’ve been labelled as having Chronic Inflammatory Response Syndrome (CIRS) before, and if you know me, I think they’re model is highly flawed and “overfitting the data”.
Don’t get me wrong I’ve spent like $20k on CIRS practitioners before, but after learning this stuff, it made me realize they looks at this stuff backwards.
I’ve even had Shoemaker telling me to cut out my gallbladder, because I didn’t tolerate VIP well LOLOL!
I still have my gallbladder, thank goodness.
Anyway back to HIF1a and HIF2a, these are adaptive responses to anaerobia, to keep you alive and breathing. R R
For example, the Tibetans (who descended from the Denisovans), are able to breath at such high altitudes because they have stronger HIF genes. R
When this adaptation response happens, you get things like Erythropoietin (EPO) and Vascular Endothelial Growth Factor (VEGF), which is favorable for improving oxygen supply to hypoxic regions. R R
In fact, HIFs upregulate stem cell factor expression, which recruits tumor-infiltrating mast cells and increases levels of cytokines Interleukin-10 (IL-10) and Transforming Growth Factor-beta 1 (TGFB1) as an anti-inflammatory effect. R R
Every tested for all of these?
I have.
Anyway, this results in an immunosuppressive (secondary infection and tumor-favorable) microenvironment. R R
In the next post, we will discuss this in the concept of micro sepsis and immune paralysis.