Junction Dysfunction Basics
I’d argue a lot of common health issues are due to something I’ve coined as “Junction Dysfunction” (JD).
JD consists of Transient Capillary Leak Syndrome (TCLS) and Micro-Sepsis (MSS) and in this post we will be discussing the latter: MSS.
Sickness Behavior and Microsepsis
When I first learned about sepsis, it was from the context of Sickness Behavior trying to learn how to improve my fatigue and brain fog.
Sickness Behavior is the model that is used to create the context of sub-lethal sepsis in animals, which causes them to initiate a whole inflammatory cascade via toll like receptors.
Anyway after learning about full blown sepsis (being on your deathbed), and the micromodels of sepsis (microsepsis/sickness behavior), it helped link those dots.
Why isn’t this sepsis? - well you're not on your deathbed, so it falls under the threshold of getting diagnosed.
In this post, I argue that microsepsis (aka sickness behavior) is a major chronic factor in Junction Dysfunction.
What Is Sepsis?
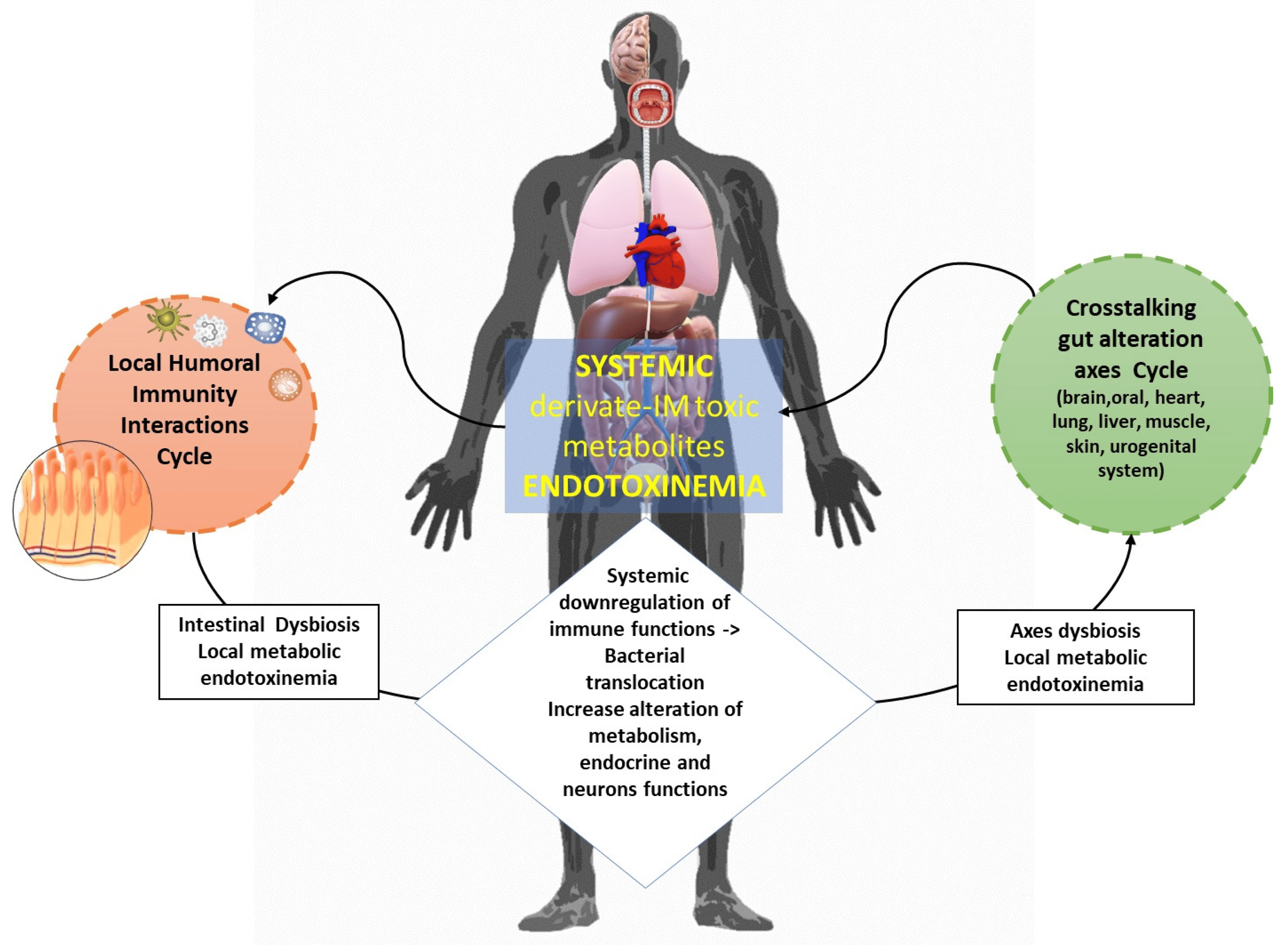
Sepsis is a serious condition where the body's response to an infection goes out of control, leading to severe organ dysfunction. R
Sepsis can affect all organs in the body - this means that it affects your brain, liver, kidneys, thyroid, ovaries, etc. R
The cascade in sepsis happens because the body's immune system first becomes very inflamed (acute innate immune activation) and then becomes weak (chronic immunosuppression), leaving patients vulnerable to acquiring other infections. R
People with sepsis often die when they are in a prolonged weak immune state, making them more likely to get new infections in the weeks or months after the initial illness. R
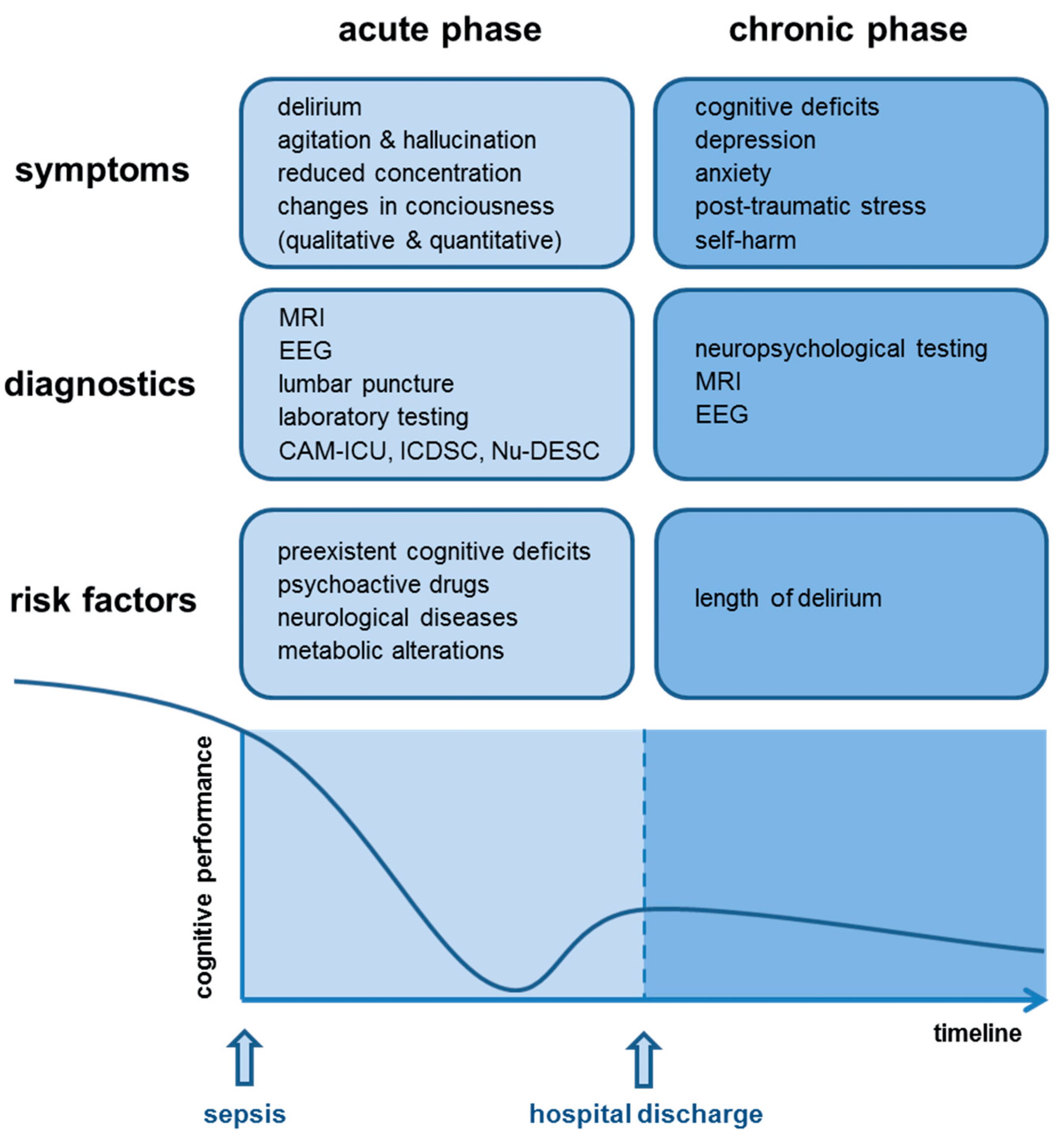
This weakened state, called "immune paralysis," is mainly caused by immune cells dying off uncontrollably (from Neutrophil Tolerance and Endotoxin Tolerance). R
This immune paralysis is what I see happening in microsepsis (more on this in the next section). R
Specifically, the death of immune cells like lymphocytes, which are important for both the initial and long-term defense against infections, increases the risk of new infections and poor outcomes. R
Studies have shown that this immune cell death happens in people of all ages, from newborns to adults, and is a major reason why sepsis leads to a weakened immune system. R
Old Models of Sepsis
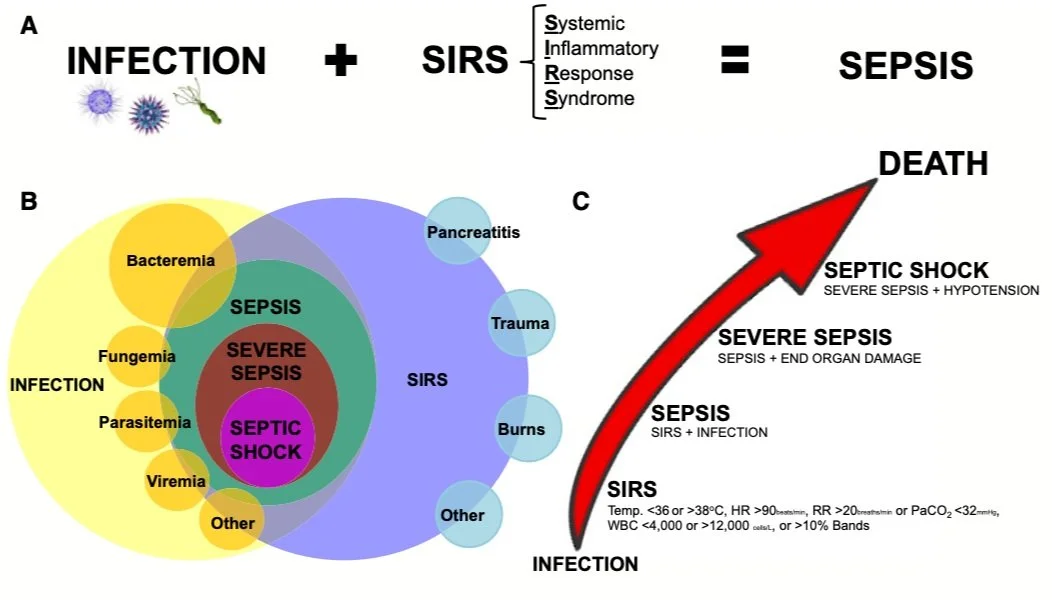
Old definition of sepsis: (Figure Above) R
the constellation of symptoms occurring when a bacterial, viral, or fungal infection leads to a systemic inflammatory response syndrome (SIRS)
fever
leukocytosis, or leukopenia
decreased vascular resistance frequently leading to hypotension (septic shock)
organ failure (severe sepsis)
death
New Model of Sepsis as Microvascular Dysfunction
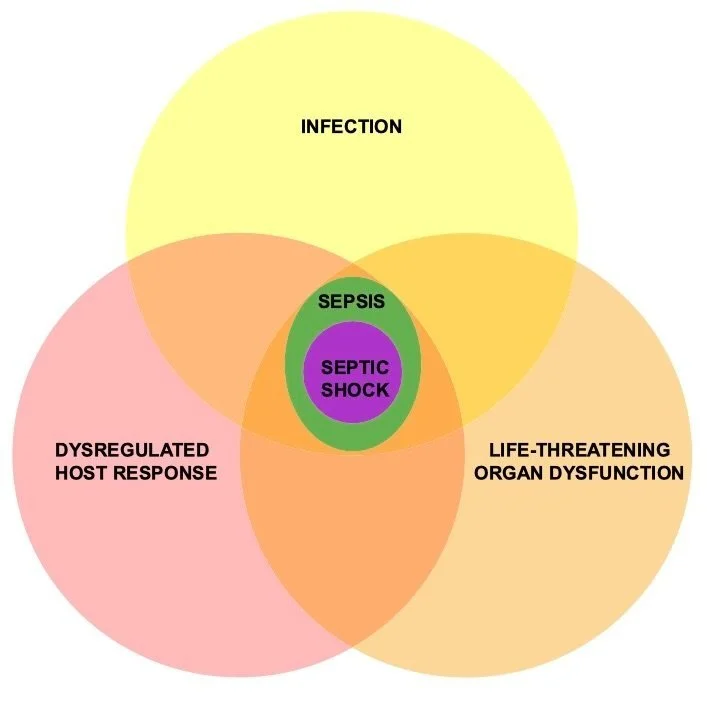
Even though sepsis involves a complicated mix of immune responses, the main driving force behind the condition is recognized as microvascular dysfunction. R
This dysfunction originates from the breakdown of the endothelial barrier, the layer of cells that line blood vessels. R
When the body responds to infection or injury, inflammation is a necessary part of the healing process. R
However, this inflammation disrupts the normal functioning of blood vessels. R
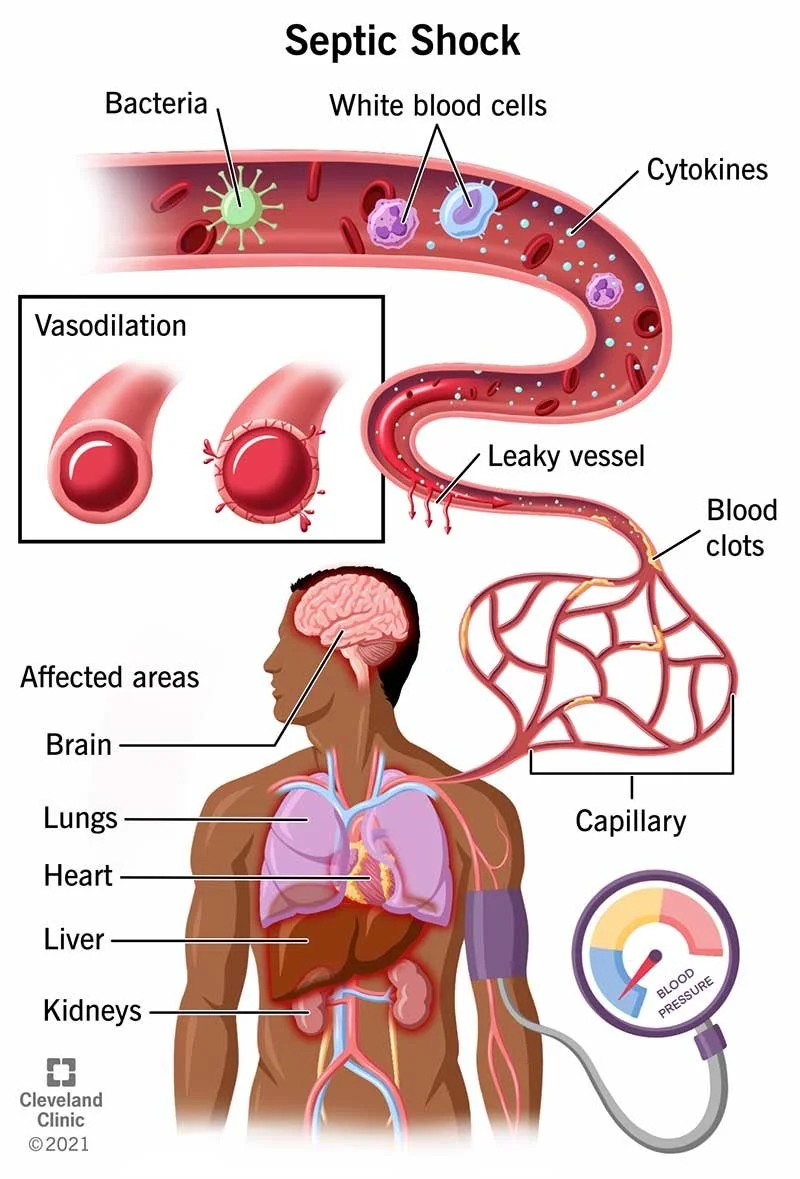
Microvessels, which are tiny blood vessels, widen, and their endothelial linings—the glycocalyx or endothelial surface layer (ESL) that touches the blood—become leaky. R
This allows fluids, substances, white blood cells like neutrophils, and macrophages to escape into surrounding tissues. R
At a local level, these "leaky vessels" might not significantly impact the overall volume of blood circulation. R
But when this process is widespread throughout the body, it can lead to circulatory failure. R
The damaged endothelium also becomes prone to blood clot formation, which increases the risk of tiny areas of reduced blood flow known as microvascular ischemia. R
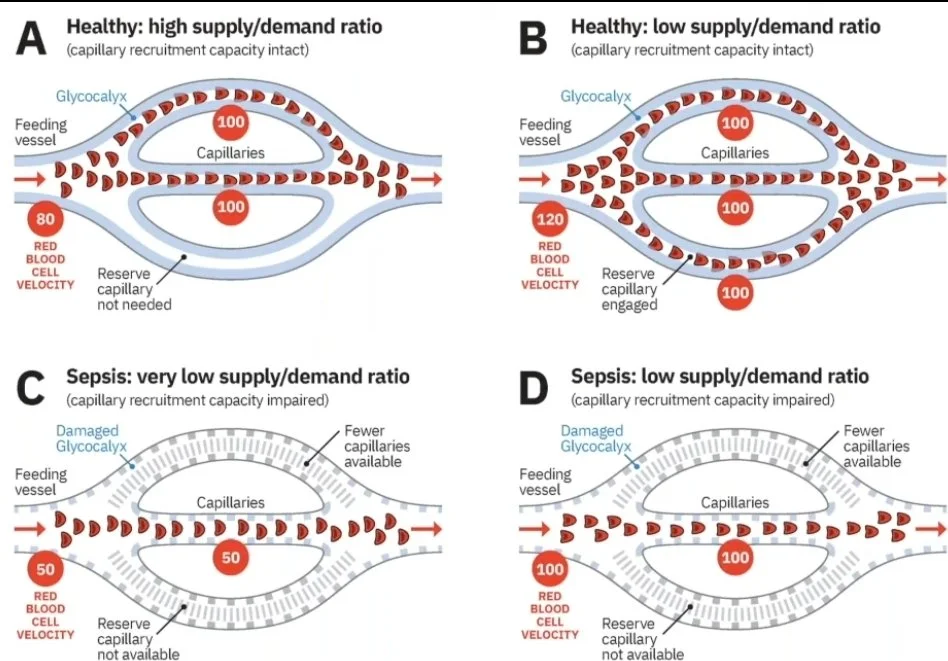
When combined with low blood volume (hypovolemia), these ischemic capillary beds cause damage to organs.
This organ damage is a major reason for the severe health problems and high risk of death seen in people with sepsis.
How Microvascular Dysfunction Expands to Other Tissue
Your microvascular system goes everywhere.
Gut
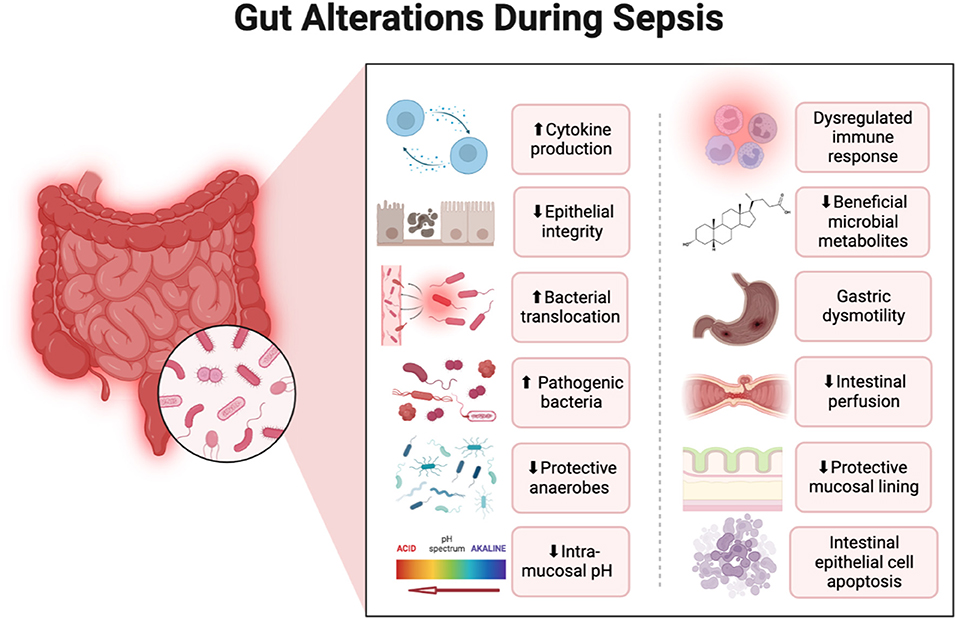
Patients with severe sepsis have reduced Bifidobacterium and Lactobacillus (two bacteria that control GABA) and increased colonization of pathogenic Staphylococcus and Pseudomonas spp. R
Sepsis induces intestinal wall permeability allowing bacterial antigens to interact with gut-resident immune cells and influence systemic immunity. R
Endotoxin released from gut bacteria exacerbate systemic inflammation and promote organ damage. R
Brain
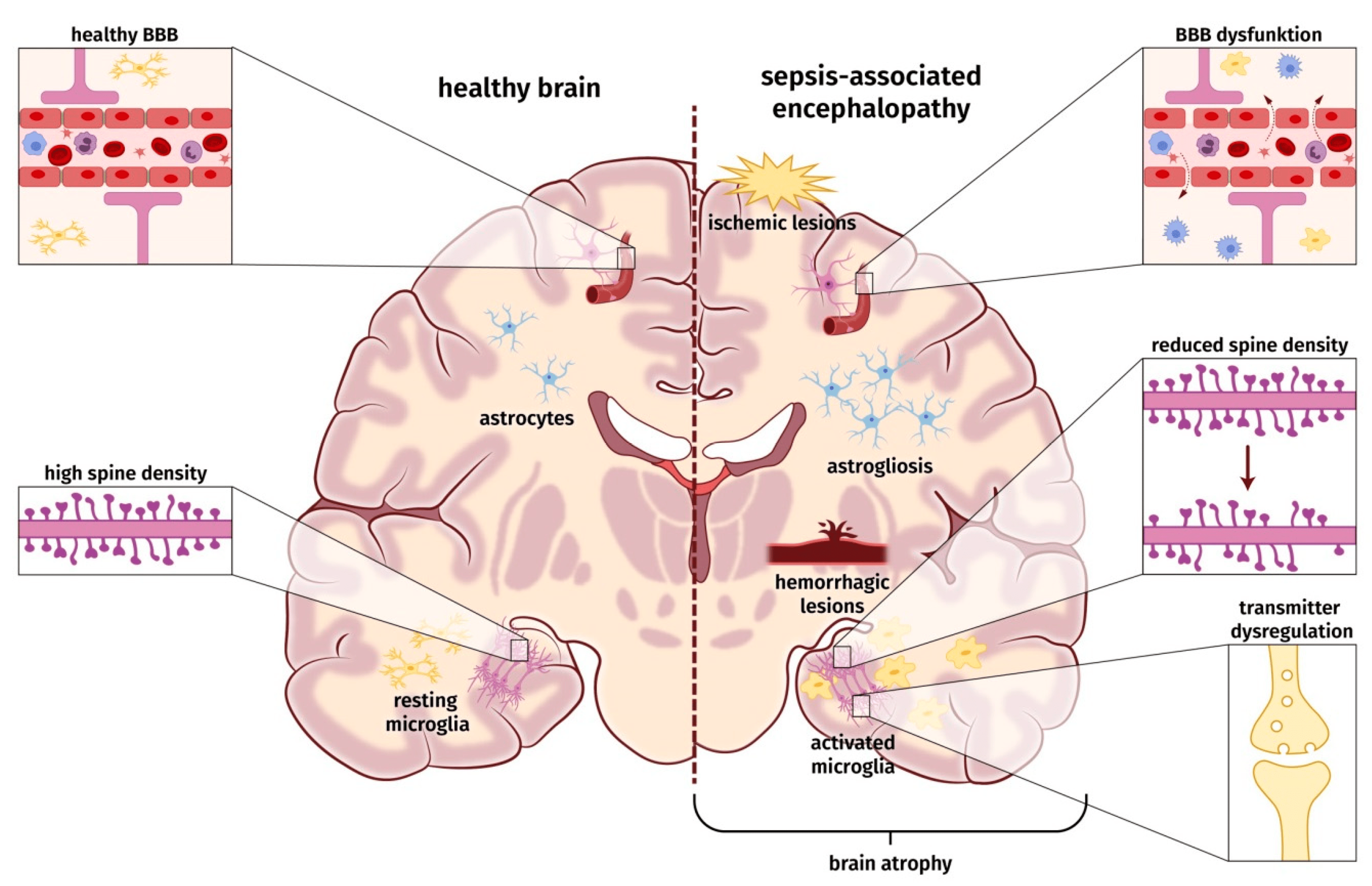
In the brain there are vascular changes include blood–brain barrier dysfunction, neurovascular uncoupling, and strokes. R
In terms of glia and astrocytes, neuroinflammation includes microglial and astrocytic activation enhancing excitotoxicity and metabolic imbalance inducing neuronal cell death. R
Mood Systems
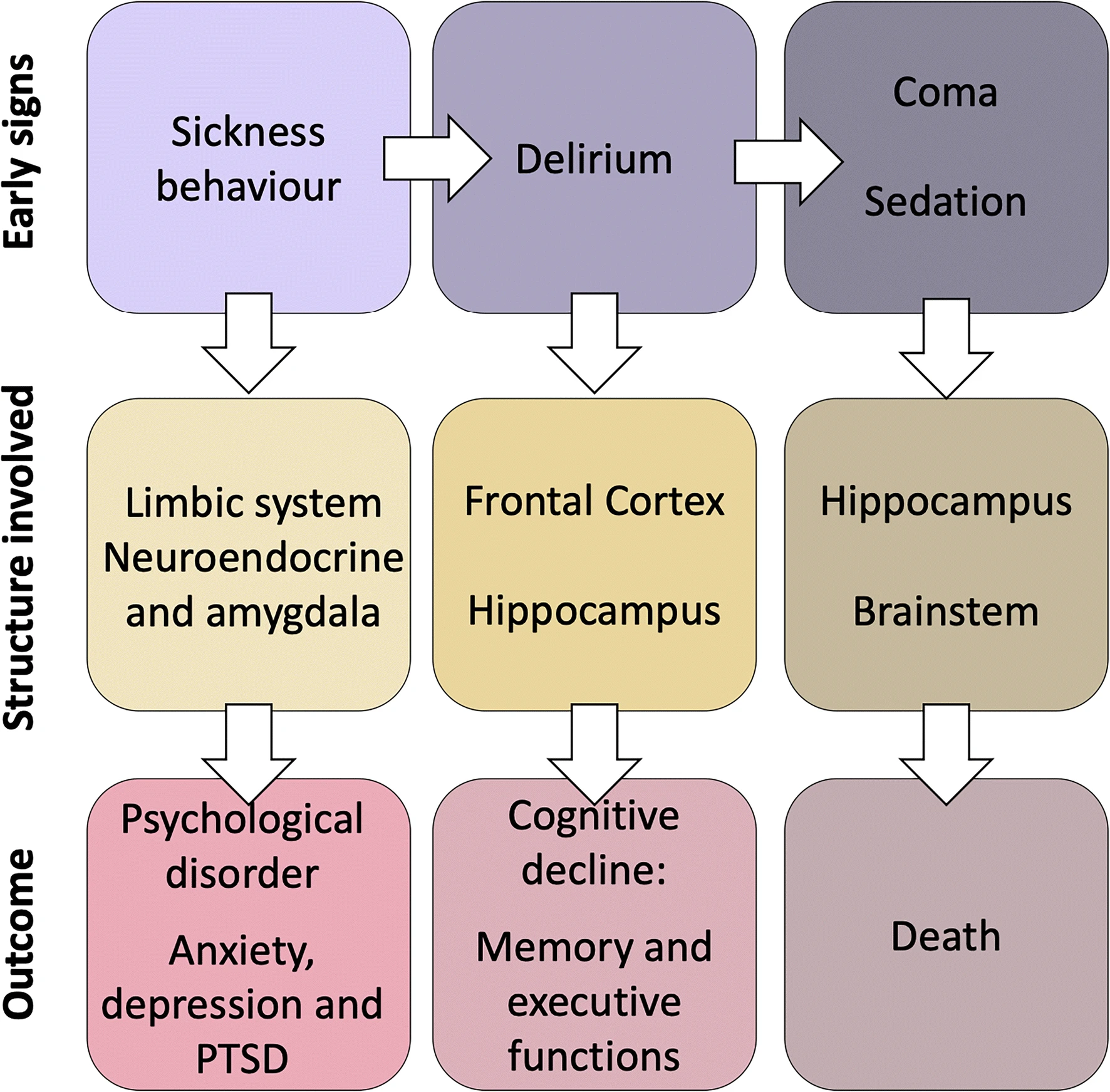
Chronic activation on sub-lethal levels cause issues with:
limbic system - emotion and sensory regulation such as anxiety, depression, and PTSD R
neuroendocrine system (HPA-axis) - autonomic nervous systems and hormones R
frontal cortex - cognitive decline R
Autonomic Regulation
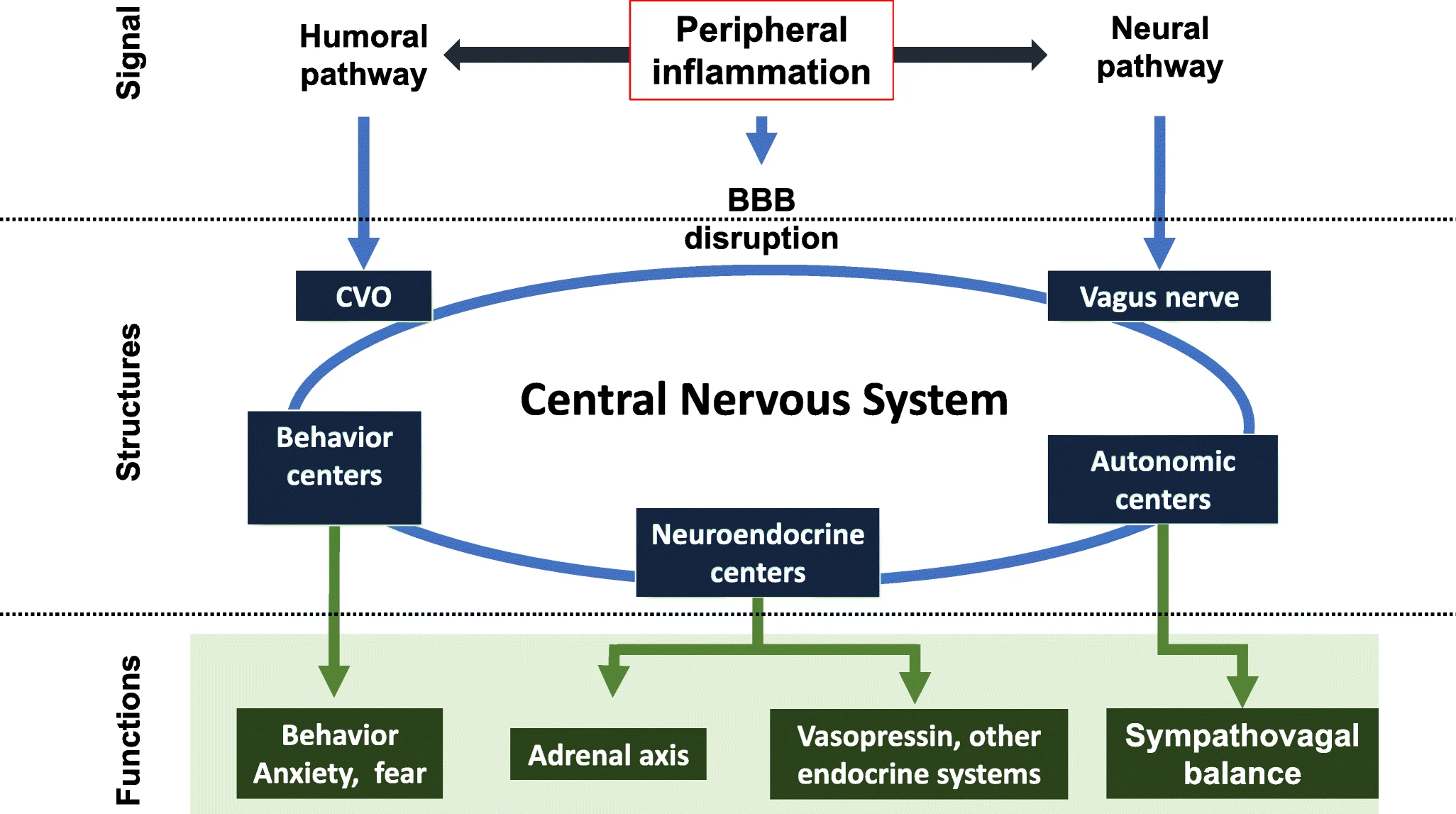
Through the neural pathway, the vagus nerve activates early the nucleus tractus solitarii and the locus coeruleus, which activate compensatory mechanisms in sepsis such as the control of blood pressure, heart rate, and arousal. R
These neural centers act as entry points to the CNS and stimulate the other autonomic nuclei and the behavioral and neuroendocrine centers. R
The precocity of the CNS activation by the neural route might explain that sickness behavior is one of the earliest features of sepsis. R
Liver
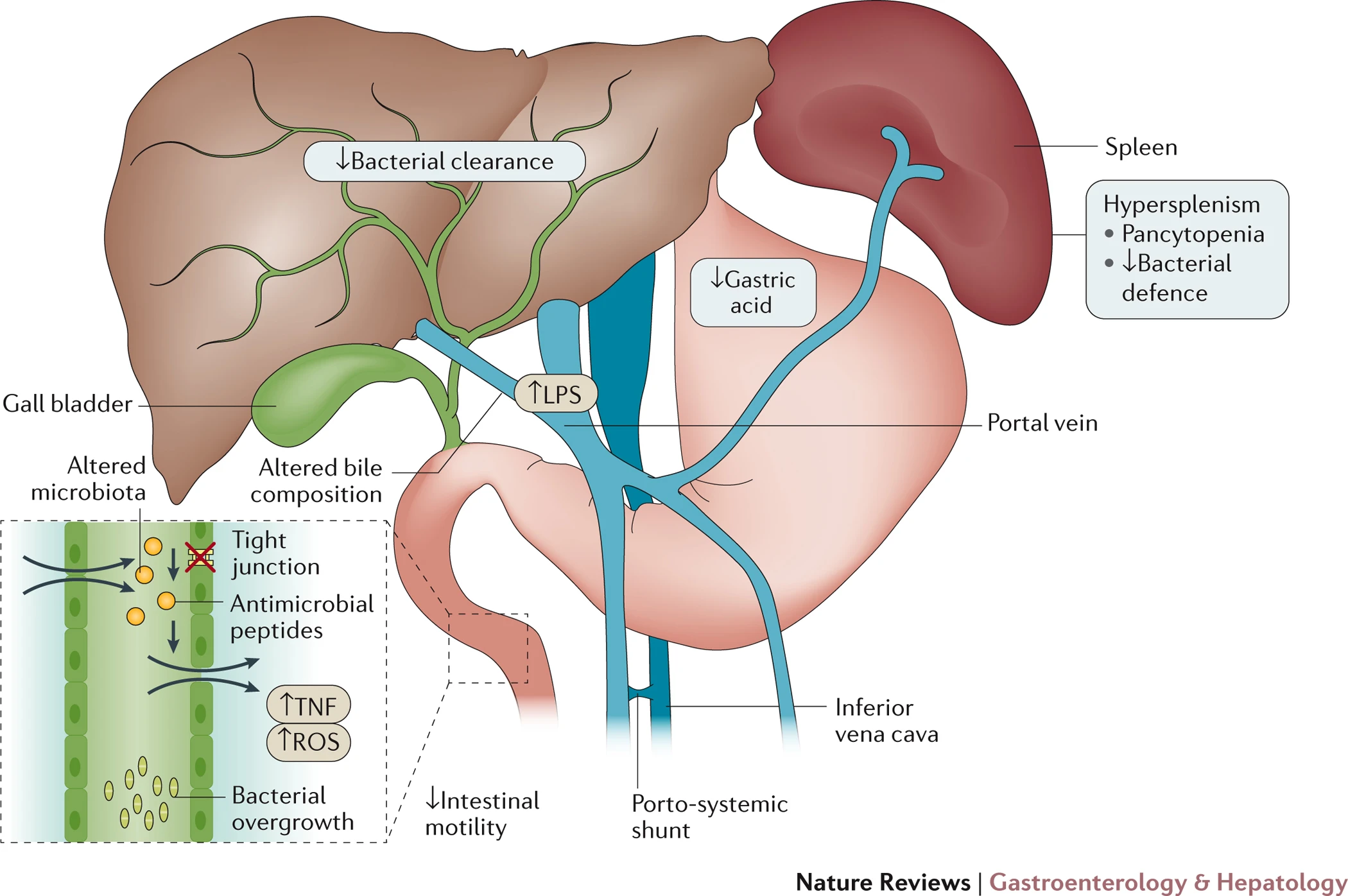
The liver represents a key integrator of microbial responses: R
During microbial infection, the liver switches from tolerogenic towards immunogenic responses and initiates the production of acute-phase proteins
Overwhelming inflammatory responses contribute to development of liver injury in sepsis and to progression of acute-on-chronic liver failure in patients with liver cirrhosis
Sepsis-induced liver injury comprises hypoxic hepatitis, sepsis-induced cholestasis and secondary sclerosing cholangitis of critically ill patients
The lowered microbial clearance allows for gallstone formation and a “sluggish” liver. R
Lungs
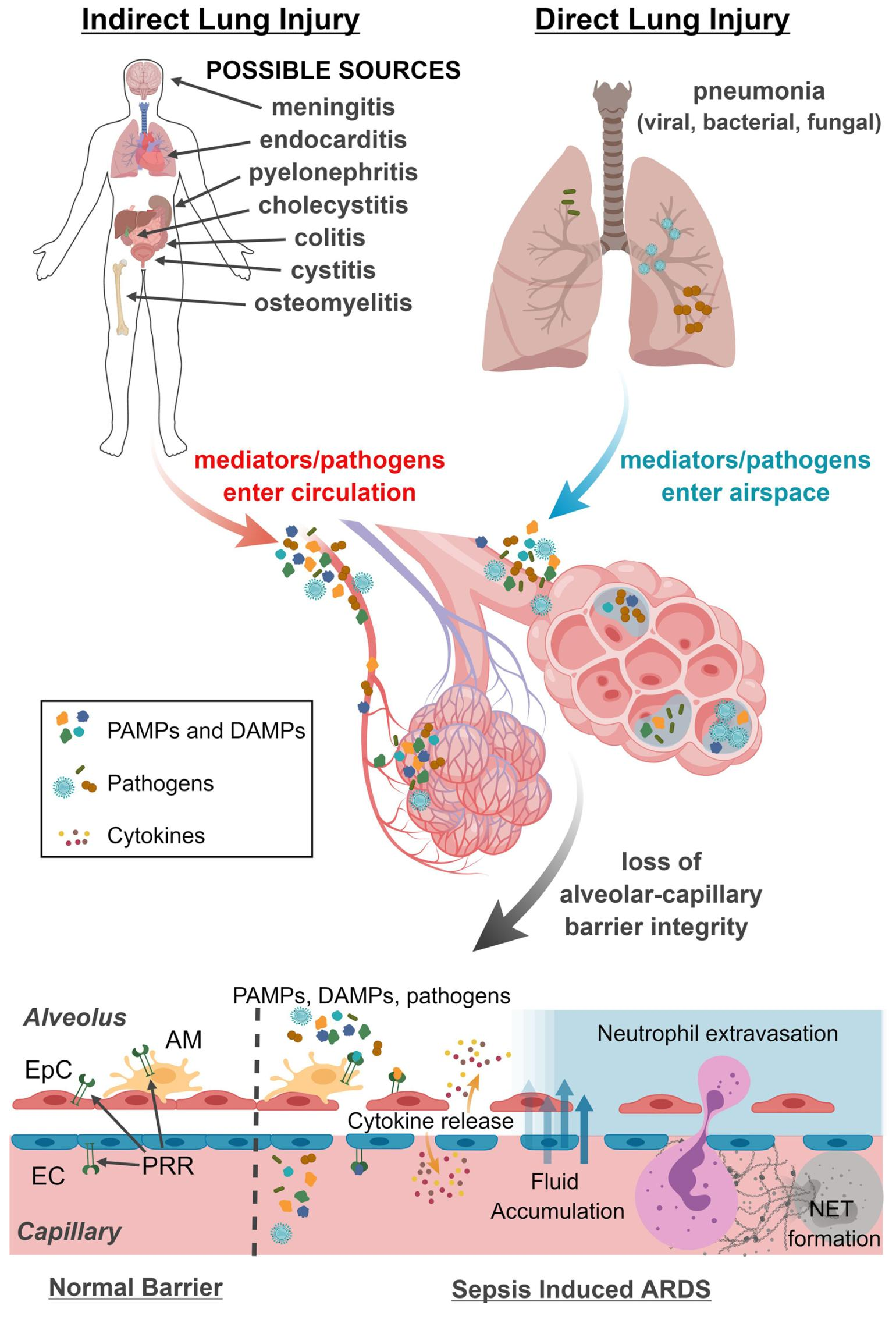
Sepsis-induced injury can initiate on the epithelial side (direct lung injury) or the endothelial side (indirect lung injury). R
Barrier dysfunction from sepsis-induced ARDS can arise from an infection originating in the lung (e.g., pneumonia) or from extrapulmonary infection (e.g., intraabdominal infection). R
Injury to the endothelial cell surface, through alteration of the glycocalyx, formation of platelet–leukocyte aggregates, and triggered release of inflammatory mediators, can also facilitate loss of alveolar-capillary barrier function. R
Hypoxia, Anaerobic, and Redox in sepsis
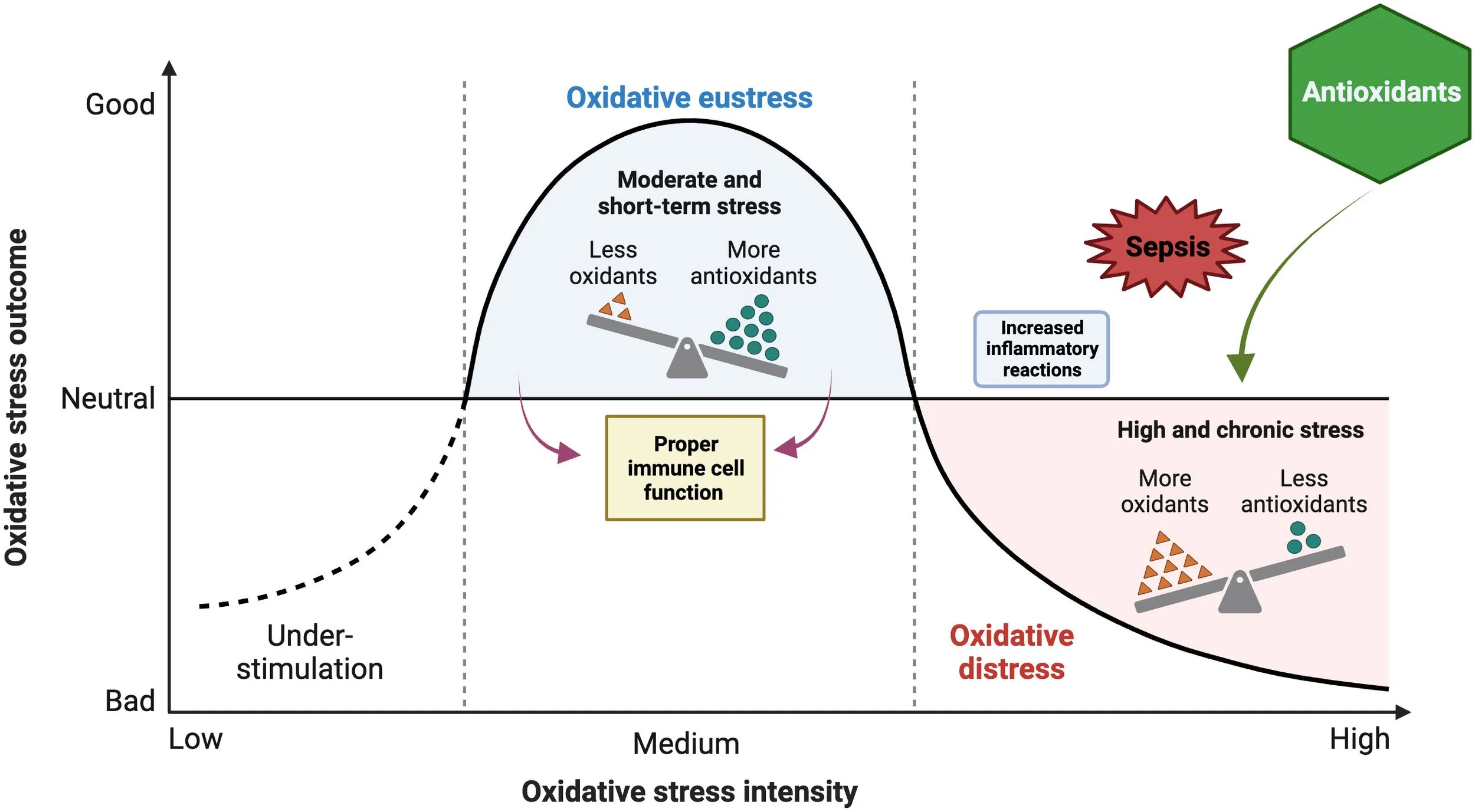
Redox
Septic patients exhibit overwhelming oxidative stress which results from uncontrolled production of reactive oxygen species (ROS) and reactive nitrogen species (RNS). R
This imbalance is produced by activated immune and epithelial cells that overexpress oxide synthases including inducible nitric oxide synthase (iNOS), or via mitochondrial production of ROS. R
Ca2
Hypocalcemia is common in sepsis and correlates with disease-scores during critical illness. R
Although systemic Ca2+ levels are reduced during sepsis, there are increased Ca2+ cytosolic levels which may stem from increased uptake. R
Heightened intracellular calcium leads to elevated inflammatory responses, cellular dysfunction, and can even be cytotoxic. R
In addition, the accumulation of Ca2+ in organs during sepsis is also associated with significant organ dysfunction. R
Hypoxia
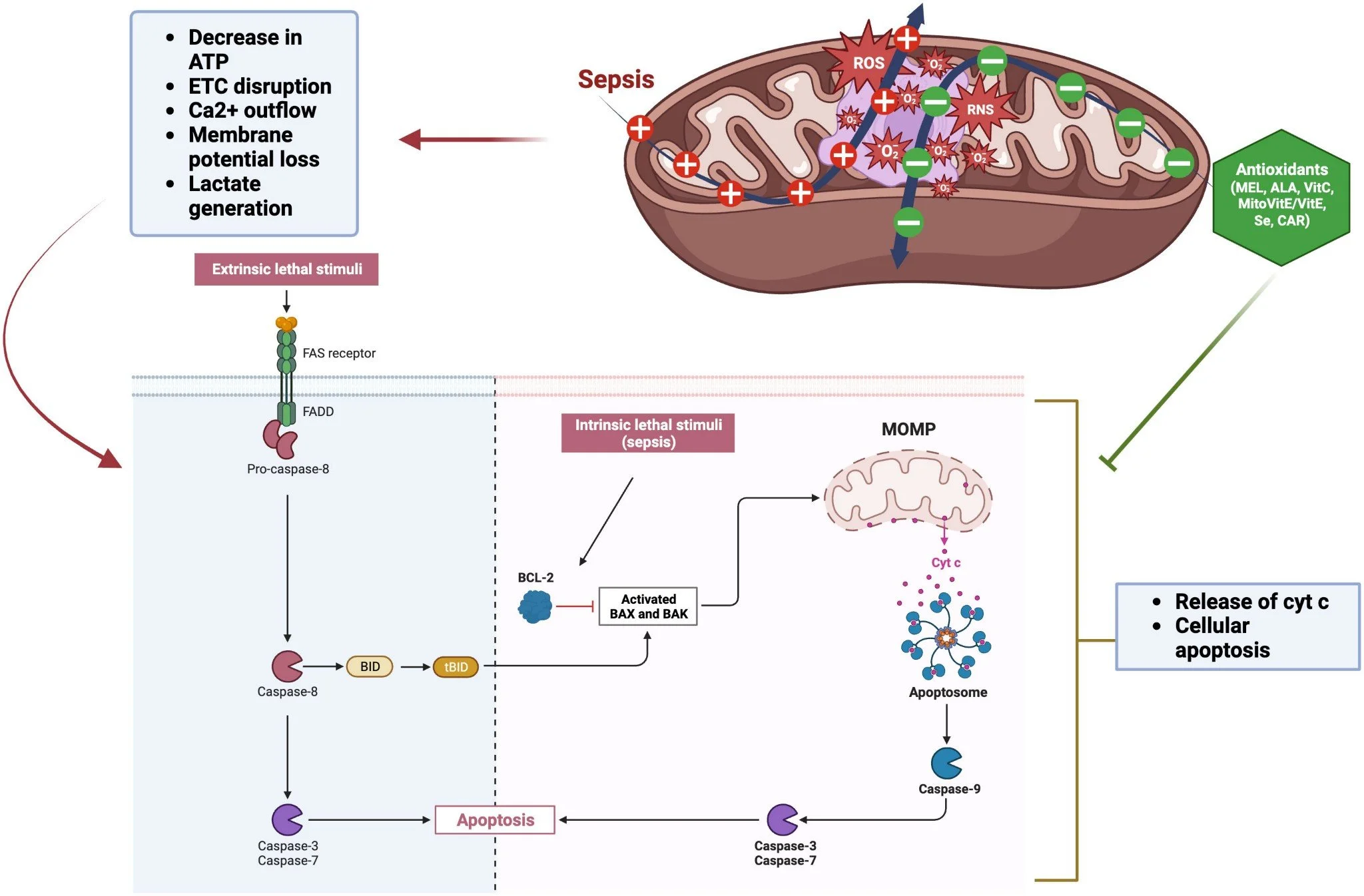
Healthy mitochondria (M2) rely on oxidative phosphoylation (aerobic mechanism). R
Fighting an infection causes HIFs to be produced and mitochondria go into a M1 phase.
Impaired tissue perfusion (due to fluid loss, both intrinsic and extrinsic, as well as reduced vascular tone) leads to tissue hypoxia.
Loss of tissue oxygenation significantly impairs oxidative phosphorylation and may trigger cell death pathways.
Acute Immune Activation, Then Chronic Suppression
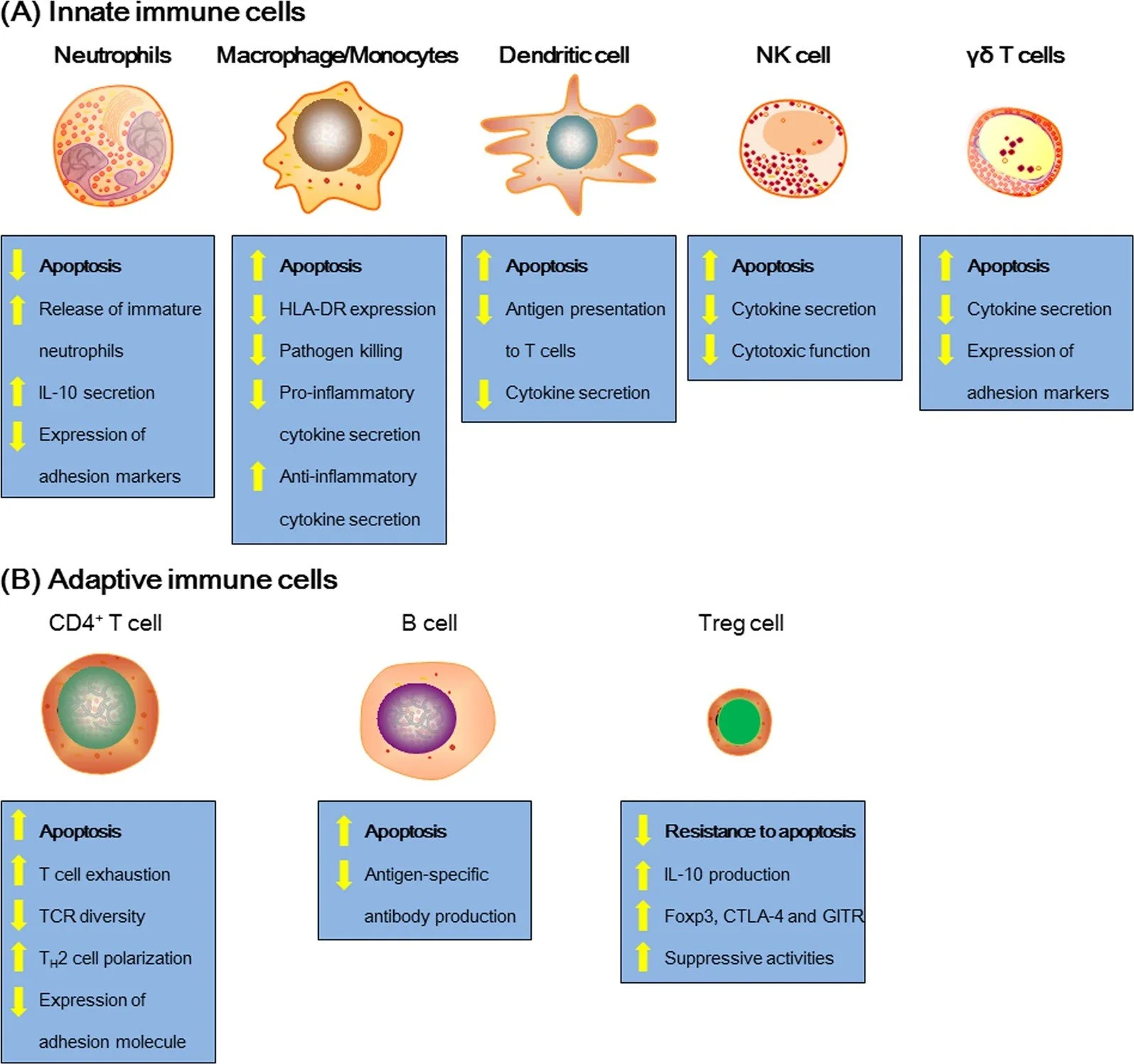
Neutrophils and NETs
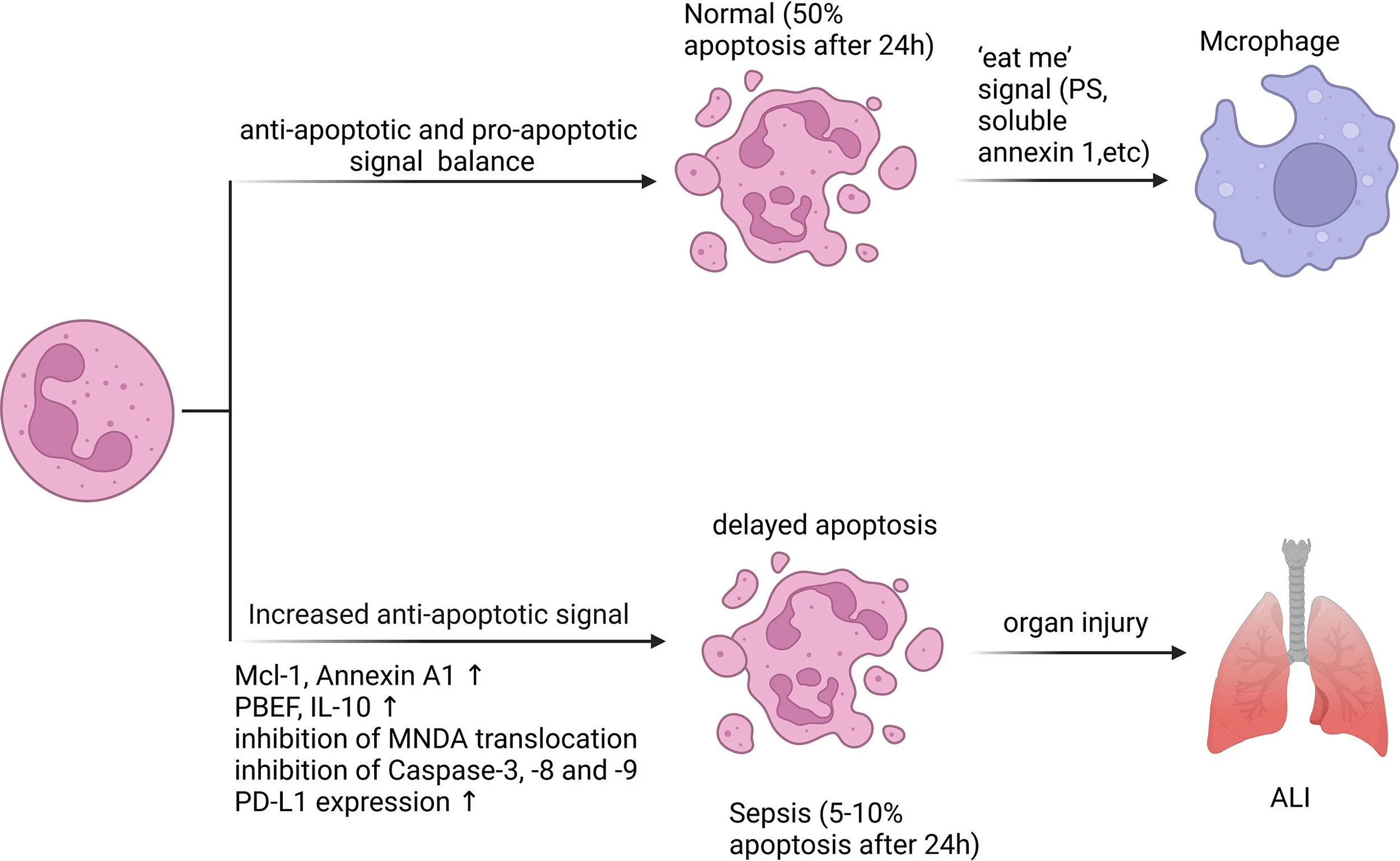
The level of neutrophils increase rapidly owing to delayed neutrophil apoptosis, as evidenced by the high neutrophil count observed in animals and sepsis patients only during the first 24 h of sepsis initiation. R
This then leads to persistent neutrophil dysfunction (neutrophil tolerance and exhaustion), compounded by the release of immature neutrophils from the BM that culminates in neutrophil deficits in oxidative burst, cell migration, complement activation, and bacterial clearance, all of which contribute to immune dysfunction and persistent inflammation. R
Monocytes and Macrophages
The most notable change during sepsis is that the impairment of bold monocytes from the patients initiates “endotoxin tolerance” and leads to poor outcome. R
Macrophages are essential in maintaining and activating host inflammatory responses. R
Macrophage phagocytosis leads to an imbalance of pro- and anti-inflammatory cytokines, resulting in polarization of macrophages to an M2 phenotype. R
Dendritic Cells
DCs link the innate and adaptive immunity. R
One way they do this is by increasing the expression of MHC class II antigen and costimulatory molecules CD80 and CD863. R
Natural Killer Cells
Under normal conditions, NK cells can induce a rapid, non-specific innate immune response against intracellular bacteria, pyogenic bacteria, fungi, protozoa, and viral infections through direct cytotoxicity against virus-infected cells and early production of cytokines that can inhibit viral replication. R
Further, NK cells are the major producers of IFN-γ. R
The most important change is that the accelerated apoptosis of NK cells leads to the decreased number of circulating NK cells that lasts for several weeks during sepsis. R
More importantly, the cytotoxic function of NK cell is decreased, which contributes to sepsis-induced immunosuppression. R
Furthermore, the decreased level of IFN-γ caused by excessive apoptosis of NK cells increases the risk of secondary infection.R
B Cells
Endotoxemia causes a transient depletion of memory B cells and regulatory B cells from the circulation, but the functional capacity of B cells to produce IL-10 is maintained. R
Gamma delta T Cells
γδ T cells are activated with increased surface expression of CD69 and HLA-DR, but the number of circulating γδ T cells is significantly lower than that of healthy subjects. R
This may be due to apoptosis-induced loss of γδ T cells in the intestinal mucosa results in higher susceptibility to secondary infections as pathogens invade the circulation or peritoneal cavity. R
Tregs
Treg cells are detrimental to the proliferation and functional activity of effector T cells and other immune cells; for example, Treg cells inhibit both monocyte and neutrophil function. R
Increased Treg cell population would prevent recovery of the immune system from excessive immune responses. R
Importantly, these resistant effects are possibly due to TLR4 deficiency and associated NF-κB signal pathways. R
Mast Cells and MCETs
Mast cells will produce Mast Cell Extracellular Traps (MCETs) in microbial infections and several noninfectious pathologies. R
Chronic Endotoxemia and Endotoxin Tolerance
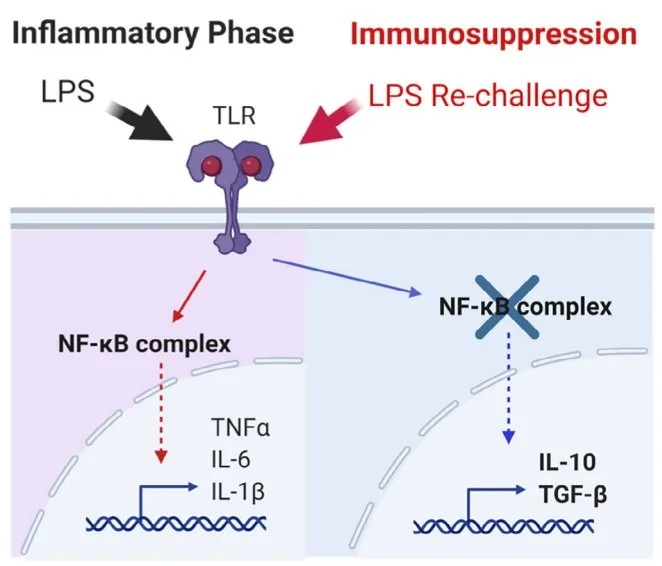
Endotoxin tolerance is a hallmark of the late phase of sepsis, where the macrophages and other immune cells experience a shift from a pro-inflammatory phenotype towards an anti-inflammatory phenotype.
Thus, cells such as macrophages secrete less TNF and IL-1β, but more IL-10.
Viruses like sars-cov-2 can also induce endotoxin tolerance (due to the sepsis mechanisms). R R
Endotoxin tolerance makes cells hypoxic (hif1a and mtor) and downrgulation of glut1.
That's probably why for those who have chronic infections do better with lower carbs and can also be thin at the same time. R
It also downregulates TLR4/MyD88, thus making it easier to acquire new infections. R
That’s why you see many people fall into the categories of CIRS and Lyme…
Sepsis, CIRS, and Lyme
So with immunotolerance to the innate immune system, also comes immunosuppression.
The immunosuppression allows for secondary infections to be acquired, such as vectorborne ones like Lyme.
These could either be new secondary infections or latent co-infections, like Bartonella. R
In those with CIRS, they have biomarkers of SIRS, which is where their model first started.
Cell Death and Autophagy
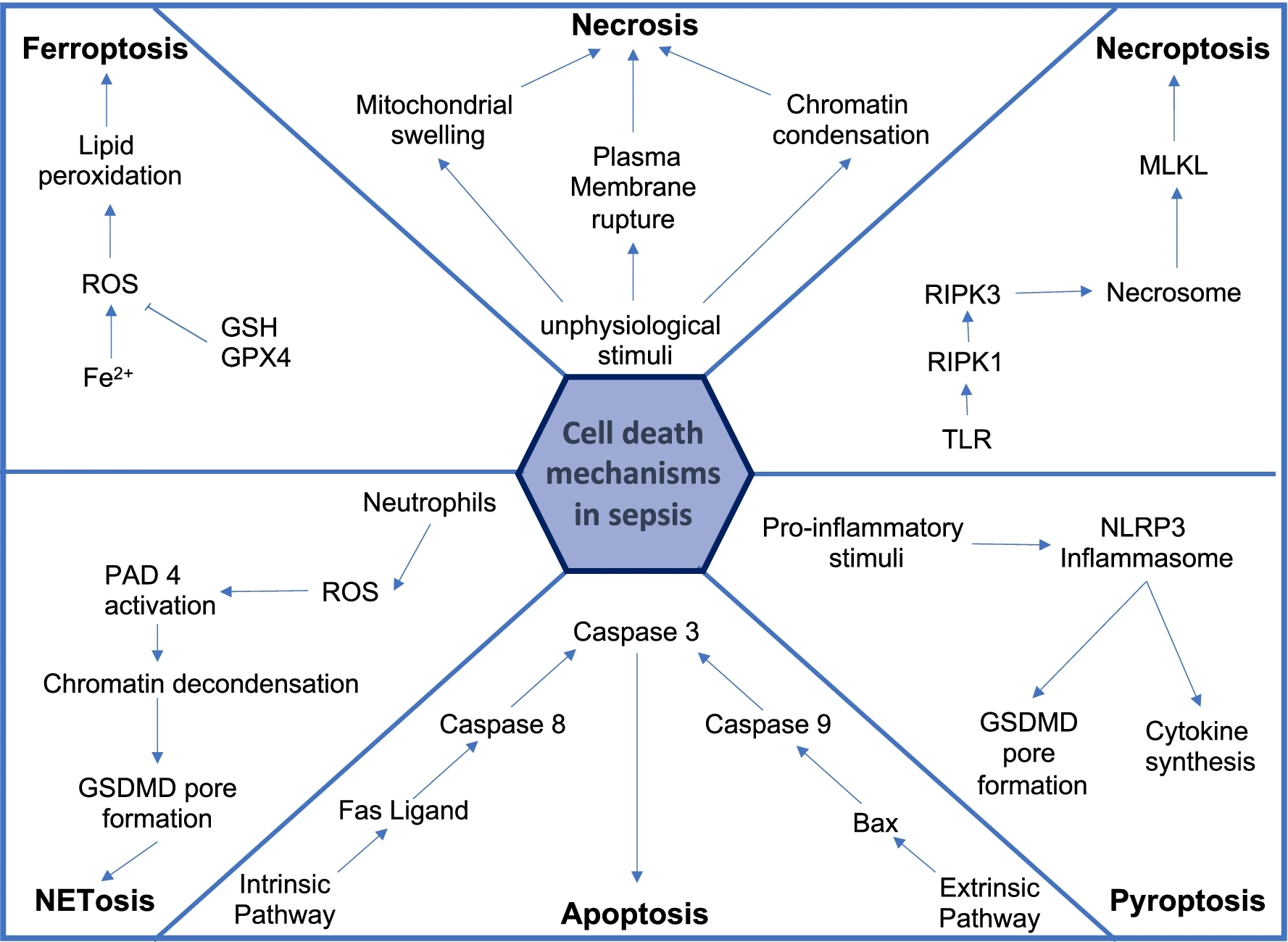
Autophagy provides a way to eliminate DAMPs and PAMPs by packaging pathogen components, damaged organelles, and cellular proteins into vesicles targeted for lysosomal degradation, resulting in reduced inflammation and cellular activation.
The problem comes with neutrophils. R
Neutrophils are key effectors of the innate immune response to sepsis. R
These cells promote pathogen clearance by engulfing and killing bacteria and by releasing NETs, which are networks of extracellular DNA, including nuclear chromatin with associated proteins, that may facilitate pathogen clearance. R
However, delayed neutrophil apoptosis and prolonged neutrophilic inflammation can mediate further capillary barrier disruption. R
This creates a lot of oxidative stress inflammation through pyroptosis, causes oxidation of not only the pathogen, but also you, the host. R
Instead of these neutrophils and NETs dying, they stay around.
This leads to a ton of oxidized debris that macrophages have to come in and clean up, and they eventually turn into foam cells, which are highly inflammatory.
Eventually macrophages will explode with all of the oxidized debris they have been trying to clean up. R
Both this process with neutrophils and macrophages causes the improper maintenance of cell death and immunosuppression, but it’s more like senescence. R
Enhancing autophagy may contribute to the protective effect in organs during sepsis. R
What Keeps Chronic Microsepsis and Endotoxin Tolerance
Anything immunogenic or antigenic.
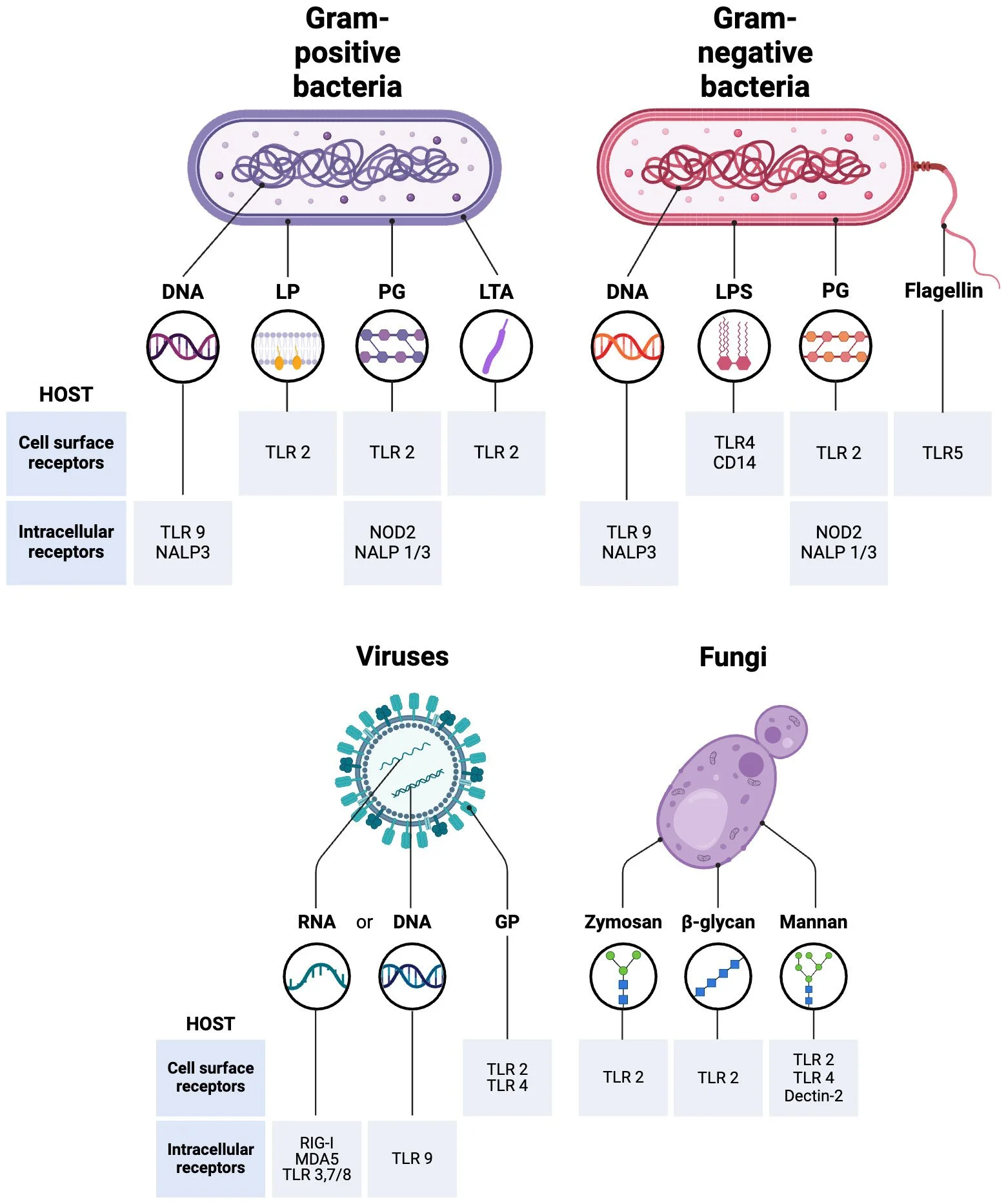
Anything that activates pattern recognition receptors (PRRs), such as MAMPs, PAMPs or DAMPs, like lectin or complement pathways, or toll like receptors.
What are examples of these?
Microbial byproducts (gram neg or gram pos bacteria, viruses, fungi) - this is why dysbiosis is a chronic factor.
It keeps endotoxin tolerance (avoid the things in the LPS post).
Toxins from the environment (or food) or from pathogens are also immunogenic and/or antigenic and can keep this cycle continuing.
Biomarkers and Testing for Microsepsis Pathologies
These 4 tests, along with the biomarkers section above, would be my ideal choices.
Functional Tests
Biomarkers
Procalcitonin (high) - Precursor of hormone calcitonin secreted by C cells of thyroid gland R R
CRP (high) - Acute phase protein secreted by hepatocytes in response to pathogen or tissue damage R
Copeptin (high) - trying to signal ADH/AVP production R
PD-L1 (high) - this is keeping neutrophils from dying R
VEGF (high) - endothelial remodeling R
HLA-DR (low) - monocytes that contain HLA-DR expression is suppressed, and thus decreases lymphocyte proliferation in response to invading pathogens. R
IL-10 (high) - Mainly produced by lymphocytes in response to infection suppresses high inflammation from the innate immune system while promoting immune tolerance via adaptive immune checking. In sepsis, IL-10 is very resistant to loss and it dampens autophagy of B cells. R
IL-6 (high) - Mainly produced by macrophages and lymphocytes in response to infection activates B and T lymphocytes R
TGF-b1 (high) - secreted by macrophages after ingesting apoptotic cells, has an anti-inflammatory property, suppressing the inflammatory innate response in sepsis. R
ADH/Vasopressin (low) - low albumin R
There are more but this is an easier list to actually test for.
In the next post, we will discuss how to get improve the microsepsis and transient capillary leak.