When Your Innate Immune System Doesn’t Work, You Must Rely on the Adaptive Immune System
When the immune system first fights something, the first line of defense is the innate immune system.
The Innate Immune System, the Glycocalyx, and Tight Junctions
Neutrophils and NETs
Neutrophils (NTs) are part of the innate immune system that are responsible for killing pathogens on the spot.
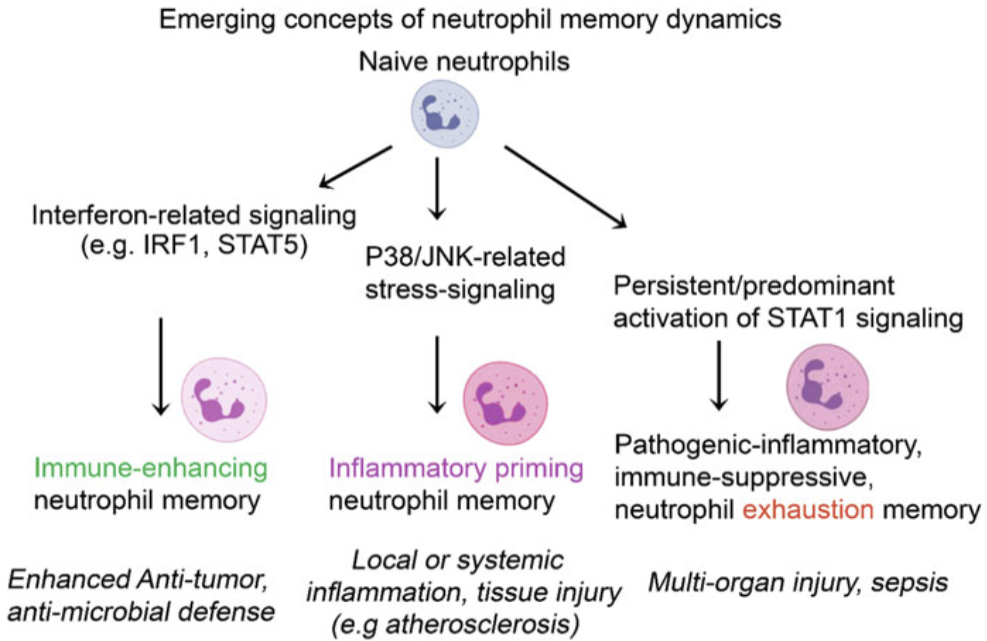
Neutrophils can be found in 3 different states/stages:
Primed - Beneficial during the infection, but detrimental in infection-free inflammation
Tolerant - Actively suppress typical immune reactions; might be advantageous in terms of constraining excessive inflammatory reactions
Exhausted - Excessive pathogenic inflammation and immunosuppression
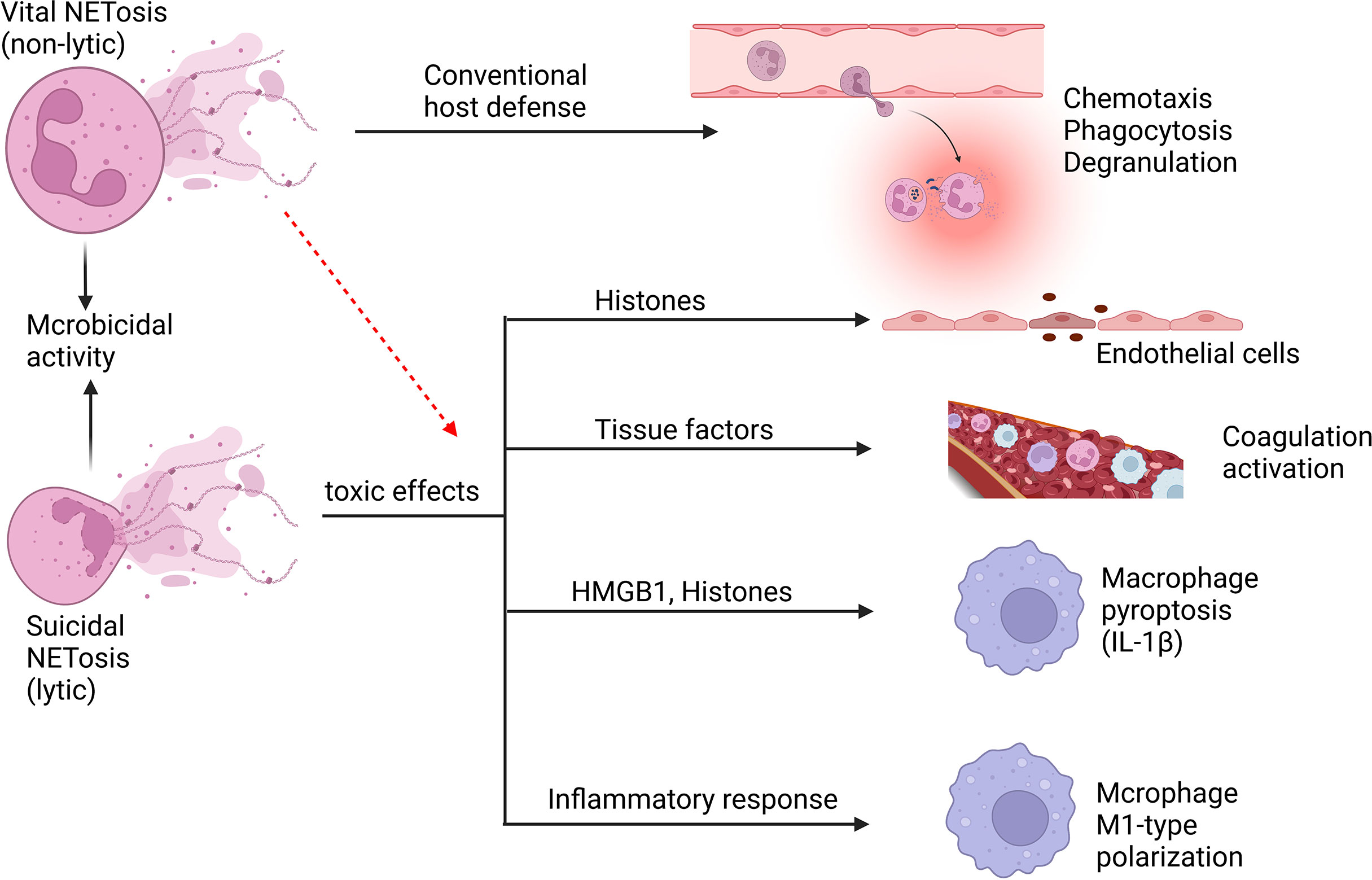
Neutrophils can release neutrophil extracellular traps (NETs) which essentially are parts of the cell (like dna) shooting out through the cell membrane.
When neutrophils release NETs in a process called ETosis, this causes pyroptosis, a very inflammatory and explosive way of cell death to keep a pathogen at bay. R
Neutrophils are constantly being formed by the bone and go to the site of infection and this creates a positive feedback loop for creating foam cells.
Macrophages
Macrophages’ job are to clean/eat up (phagocytose) debris and waste from fighting an infection (like dead neutrophils, dead bacteria, or oxidized cell components).
Macrophages can be pro-inflammatory (M1) or anti-inflammatory (M2):
M1 macrophages are good at fighting and are very inflammatory (more on this later) R
M2 macrophages are anti-inflammatory, able to do cell turnover well and regenerate efficiently R
There is a third state that is neither M1 nor M2 macrophages, but their own unique state that causes them to be both:
senescent (hanging doing nothing)
apoptotic (dying)
This state creates an immunosuppressive and highly inflammatory environment, and is usually a result of neutrophil exhaustion, endotoxin tolerance, and TNFa tolerance. R
Exhausted and Tolerant
Neutrophil Exhaustion
Constant Toll Like Receptor (TLR) activation causes a unique state in microsepsis.
After microsepsis, neutrophils can get in an exhausted state: pro-inflammatory and immunosuppressive.
Endotoxin and TNFa Tolerance
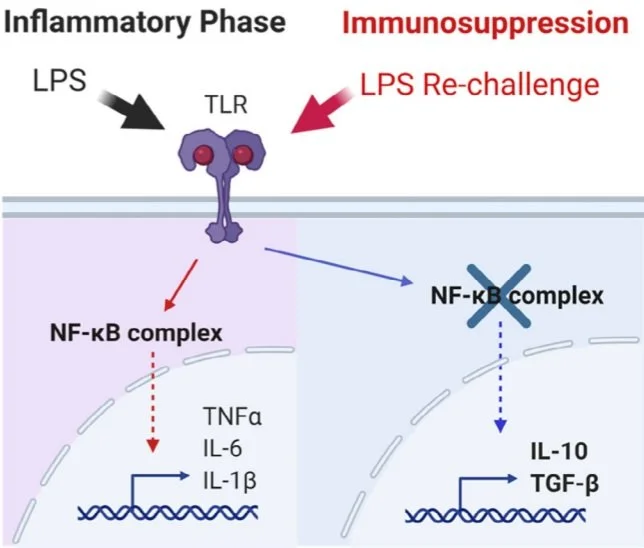
Also after microsepsis, endotoxins are present due to TCLS, and this makes macrophages constantly put in a chronic M1 state.
As an adaptive mechanism, macrophages become tolerant to chronic exposure of a pathogen.
This is a shift into a new state called Endotoxin Tolerance. R
Endotoxin Tolerance of macrophages are macrophages’ way of keeping inflammation at bay (IL-10, TGFb) as the immune system is trying is trying to protect from excessive damage while fighting (pushing macrophages into a pseudo-M2 state). R
This leads to neutrophils and macrophages becoming senescent and this creates a favorable environment for antigenic issues and cancer.
This is because your first line of defense (the innate immune system) is now suppressed:
Neutrophil Exhaustion - Neutrophils create pro-inflammatory cytokines and ROS
Endotoxin Tolerance - Macrophages create anti-inflammatory cytokines and ROS
Instead of your innate immune system doing it’s job, you have to rely on your adaptive immune system to take over as they must clean up the damage.
This is a big problem with people I’ve worked with who’ve had CIRS and I see it with Junction Dysfunction, too.
Due to having neutrophil exhaustion and endotoxin tolerance (and not so much a slow HLA tagging), it is much easier to acquire secondary infections and gain more symptoms infecting new areas of the body.
Anyway in microsepsis, similarly, TNF tolerance can lead to chronic inflammatory diseases such as rheumatoid arthritis and lupus. R
TNF tolerance may explain why TCLS goes hand in hand with limbic system disorders like low mood and depression. R
This is how you can have inflammation, immunosuppression, auto-immunity, and cancer - all in the same environment.
This environment is highly oxidative and throws off your redox balance.
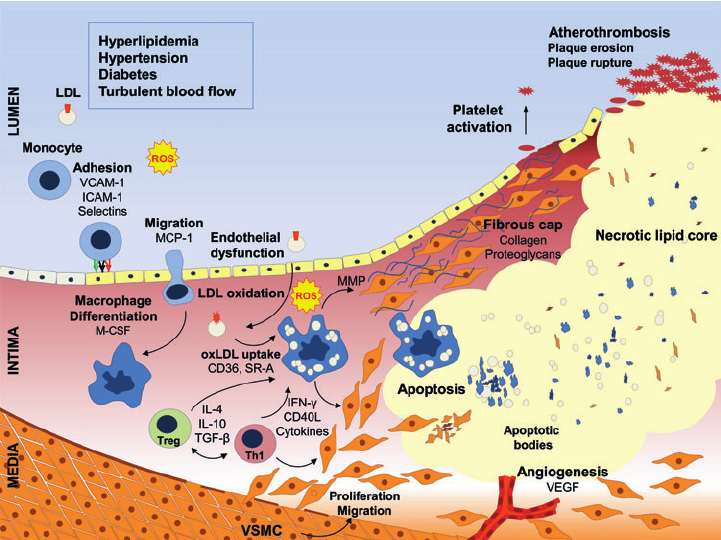
Foam Cells, Lipids, and RAGE
Foam cells are macrophages that eat up oxidized lipids, like oxidized LDL (oxLDL).
Fighting an infection makes a lot of oxidative stress (like ROS) from mitochondria in hypoxia.
This ROS that is created can kill pathogens but also hurt your own cells in the process, like plasmalogens of your lipids, or other new cellular debris.
Its macrophages job to clean up this mess and the consumption of highly oxidized lipids creates foam cells.
Foam cells will rupture (pyroptosis) eventually (when AGEs bind to RAGE on macrophages). R
This is what causes fibrotic plaque (like atherosclerosis). R
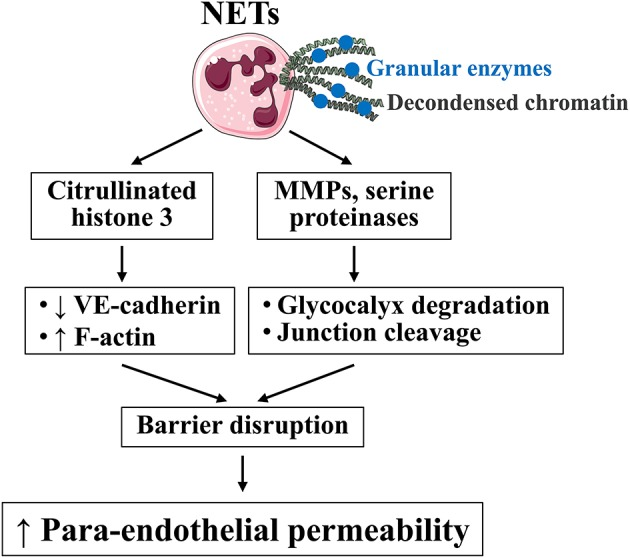
Glycocalyx Removal
Just like your endothelial system, your macrophages have a glycocalyx that helps them move freely.
Similarly, pathogens like bacteria also have a glycocalyx that allows them to float around easily as well.
In order for macrophages to capture and eat pathogens, they need to shed Metallomatrix Proteins (MMPs) and Hyaluronidases (HA-ase) to cut up their and the pathogens glycocalyx’s. R
Once they cut up their glycocalyx’s they can easily both bind to LDL, which acts as an opsonin to help phagocytosis of pathogens.
MMPs
Pathogens activate toll like receptors (TLRs) on macrophages, and this initiates the release of MMPs.
Continuous release of MMPs from TLR activation during endotoxin tolerance continues to break down the cofactors needed for the glycocalyx, and thus: R
everything sticks together
phagocytes will eat the host instead (as they have lost their selective ability)
phagocytes eat RBCs
phagocytes eating connective tissue
HA-ase
Breaking down the glycocalyx is required for macrophages to do phagocytotic activitiy.
Macrophages will use HA-ase as one enzyme for breaking down pathogens.
Unfortunately this causes collateral damage and also breaks down your collagen, so being stuck in inflammation or having constant debris needing to be phagocytozed will lead to connective tissue issues. R
Those who already have connective tissue issues will feel the effect of inflammation much more. R
Monocytes and COVID
Macrophages are derived from monocytes.
In COVID specifically, according to Bruce Patterson's research, non classical monocytes can express the fractalkine receptor CX3CR1 and bind to fractalkines on endothelial cells. R
Fractalkines are upregulated from inflammatory cytokines like IL-1, IFN-γ, and TNF-α. R
One reason why people may feel PEM after exercise or stressful events may be due to these non classical monocytes that have poorly eaten up the S1 protein of SARS-COV-2. R
Stress and exercise (4x) can mobilize non-classical monocytes by activation of platelets (see more in post about clotting and Autonomic POTS). R
Activated platelets also release CCL5/RANTES which binds to endothelial cells, promoting monocyte adhesion to inflamed endothelial tissues and acting as a chemoattractant for inflammatory cells. R
Autophagy
The key to returning senescent macrophages back to normal is through autophagy.
Autophagy is the ability for your cells to eat and recycle your own cells to be more efficient, thus cleaning up the senescent macrophages is helpful.
Ways to induce autophagy:
Lifestyle
Fasting
Ketosis
PD1
PD-L1 (or B7-H1) is a protein produced by cancer cells that interacts with PD-1 and suppresses activated T-cell from engaging with cancer cells. R
PD-L1 ligand activity may also induce apoptosis of T-cells. R
PD-L1 overexpression is observed in melanoma, pancreatic, lung, and other types of cancer cells. R
PD-L1 and it’s cognate receptor PD-1 exerts an inhibitory effect on macrophages phagocytotic activity, so it’s blocking the PDL1/PD1 pathway is important.