Post Overview
You were most likely taught that autoimmunity is your body (or immune system) attacking YOU, like as if it really wanted to hurt you.
Or maybe you were told that gluten (gliadin antibodies) is attacking your thyroid (TSH, TPO antibodies, etc).
Maybe you have high levels of ANA or AMA.
I wonder if they told you that your immune system is literally doing its clean up process after an inflammatory response…
In this post, we will discuss the inflammasome (inflammatory cascades), alarmins (antigens), and opsinins (antibodies).
Learning about how these systems work together will give you a clear perspective on how antibodies work and why “autoimmunity” term is a misnomer.
Antibody Testing
After learning about these mechanisms, I now use antibody testing in a target fashion:
Antibodies and Antigens Video
Inflammasome and Alarmins 101
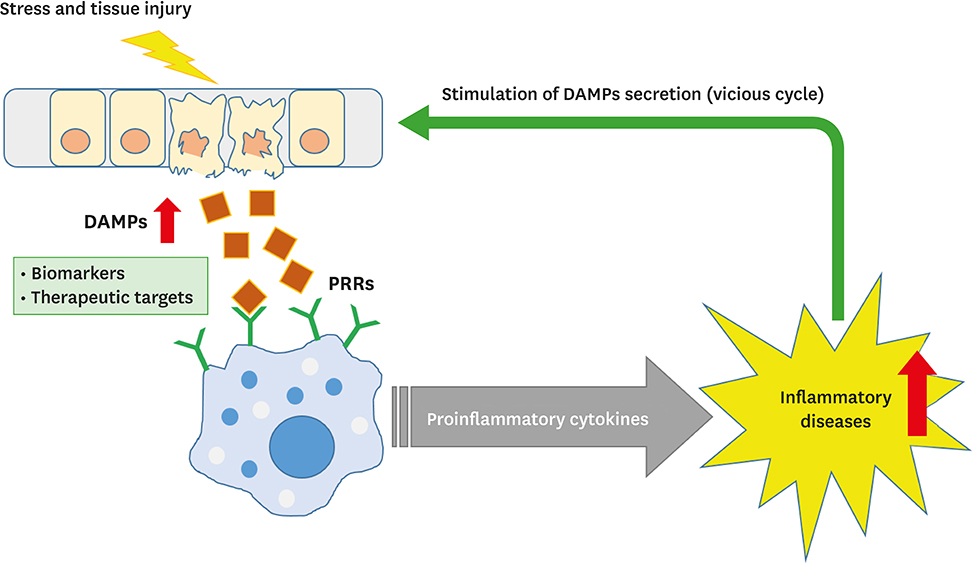
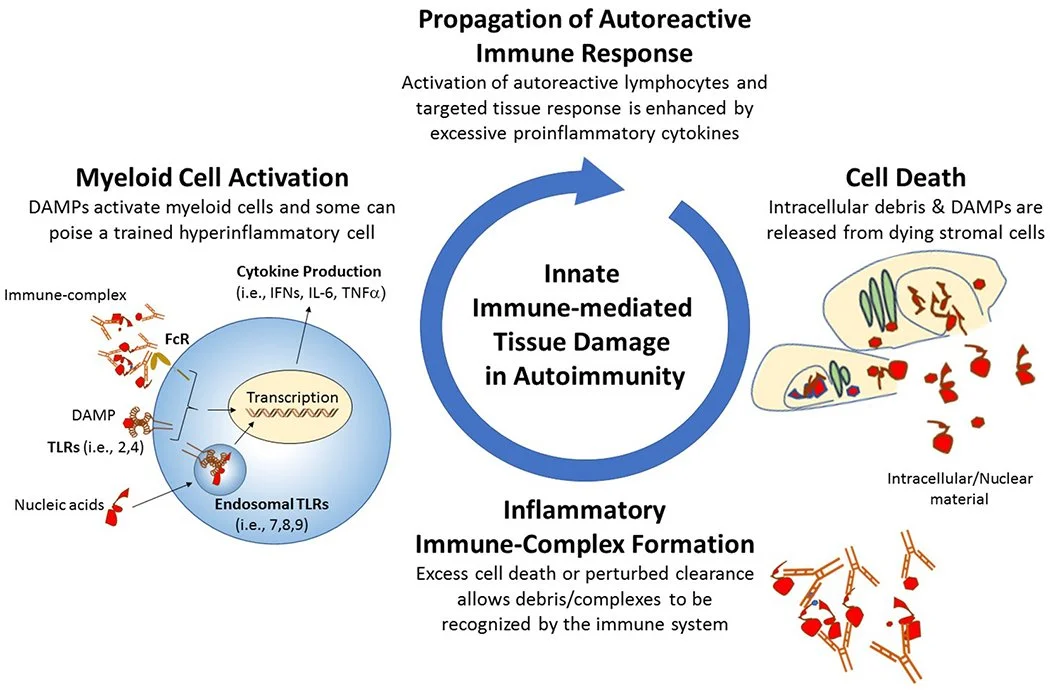
Tissue damage is a byproduct of inflammation.
When there is an infection, toxin, stressor, etc, the cells turn on what is called the inflammasome. R
The inflammasome creates inflammation and releases Alarmins. R
Alarmins turn on the inflammasome of other cells and this creates a positive feedback loop, and in some people this can become chronic. R
Alarmins are signals to the cell that there is stress or damage of a cell. R
An alarmin can be anything ranging from actual parts inside of a cell (like the nucleus, mitochondria, etc), outside the cell (like receptors), adhesion molecules, cytokines, proteopathies, and microbial parts. R
The prominent categories of alarmins are: R
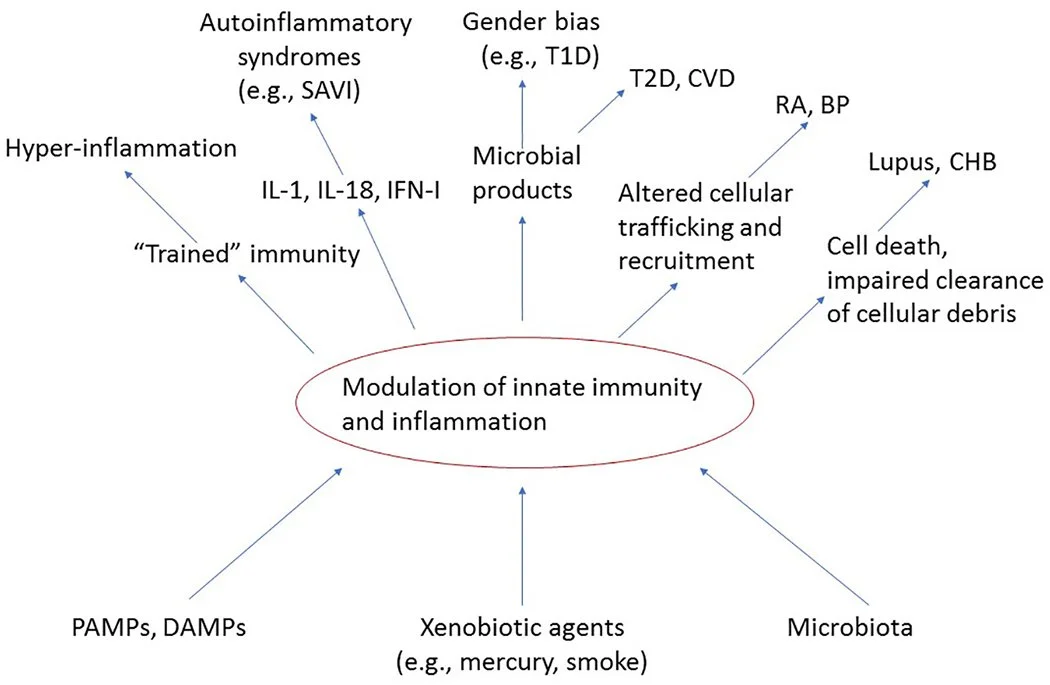
Damage/Danger-Associated Molecular Patterns (DAMPs) - released from stressed and damaged cells
Pathogen-Associated Molecules Patterns (PAMPs) - from pathogens (like LPS and Flagellin) as well as various xenobiotic agents (e.g., mercury, cigarette smoke, glyphosate and silica) R
Chromatin-Associated Molecular Patterns (CAMPs) - molecules derived from the nucleus or molecules associated with chromatin (Mitochondrial DNA, Cell-free (cf) RNAs, MicroRNAs, Telomeric repeat-containing RNA (TERRA), Extracellular traps (ETs), RNA- or DNA-binding proteins, Heat shock proteins (HSPs), Members of the S100 family, Cell free DNA (cfDNA)
Homeostasis-Altering Molecular Processes (HAMPs) - act as signal integrators that are capable of detecting perturbations in cytoplasmic homeostasis, ie - as initiated by infection R
When these alarmins are freely floating outside of the cell (in the extracellular space), this triggers the inflammasome by these alarmins binding to the Pattern-Recognition Receptors (PRRs) on the outside of cells, which have evolved to recognize stress and danger signals. R
PRRs are divided into five sub-families: R
Toll-like receptors (TLRs)
Nucleotide-Binding Oligomerization Domain (NOD)—Leucine Rich Repeats (LRR)—Containing Receptors (NLR)
Retinoic Acid-Inducible Gene 1 (RIG-1)-Like rReceptors (RLR; also known as RIG-1-like helicases—RLH)
C-Type Lectin Receptors (CLRs)
Absent in Melanoma-2-like Receptors (ALRs)
Coming up… (slightly technical)
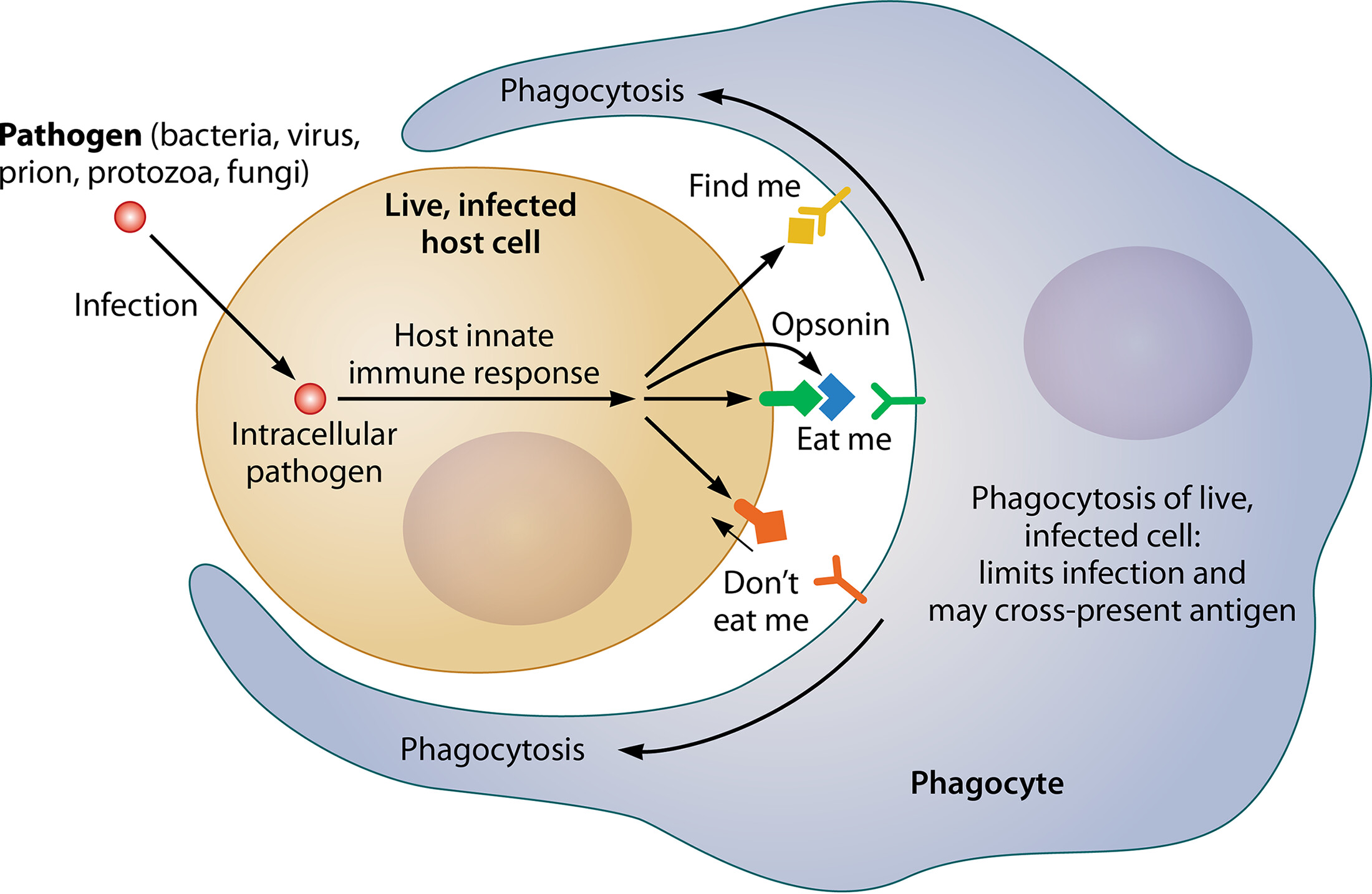
When inflammasome mediators are active, macrophages and endothelial cells release factors that attract neutrophils (NETosis) and monocytes (phagocytosis) to the site of inflammation. R
Macrophages and dendritic cells (DCs), are specialized in exposing antigens (alarmins) to lymphocytes (T and B cells), then lead to antibody (opsonin) secretion for phagocytosis of the antigens-antibody complex. R
Opsinization 101
Opsonins were discovered and named "opsonins" in 1904 by Wright and Douglas, who found that incubating bacteria with blood plasma enabled phagocytes to phagocytose (and thereby destroy) the bacteria.
They concluded that: “We have here conclusive proof that the blood fluids modify the bacteria in a manner which renders them a ready prey to the phagocytes.
We may speak of this as an “opsonic” effect (opsono - I cater for; I prepare victuals for), and we may employ the term “opsonins” to designate the elements in the blood fluids which produce this effect.
What Are Opsonins And Dysopsonins?
Cells talk through a variety of mechanisms and one of them is based on charge.
Opsonins are charged “eat me” signals that signals to the immune system to phagocytize (eat up). R
Dysopsonins are the opposite of opsonins - they signal as “don’t eat me” signals. R
Killing pathogens requires creating a lot of opsonins so immune cells can stick/bind to it and eat up that pathogen.
This is most likely an adaptive response to lots of damage needing to be fixed (more about this later when we talk about PAMPs and DAMPs).
Examples of Opsonins and Dysopsonins
Cholesterol
Cholesterol is a good example of an opsonin that the liver makes.
Cholesterol will bind to pathogens or toxic substances (like LPS or glyphosate) in order for the immune system to bind to it to eat it up.
When cells are dying they send out “eat me” signals, so macrophages can eat them up. R
For example, LDL is an eat me signal and high levels of it mean your body is crying for more autophagy. R
HDL on the other hand is an opsonin for endotoxins. R
Antibodies
Antibodies are opsonins.
For example, the γ chains of IgG are potent opsonins. R
Opsonins and their respective locations/uses:
Immunoglobulin A (IgA) - often exists in a monomeric state when in the bloodstream but it is secreted into the lumen of mucosal surfaces in a dimeric form. The dimeric form is joined together by a "J chain" with two antigen-binding sites for the neutralization of pathogens. IgA can be found in saliva, tears, respiratory, gastrointestinal (sIGA), and genitourinary secretions.
Immunoglobulin M (IgM), on the other hand, exists as a pentamer joined by a "J chain" when secreted into the bloodstream. Its response to pathogen invasion is mostly swift while that of Immunoglobulin G is delayed. When IgM is affixed on the surface of B cells, it exists as a monomer.
Immunoglobulin G (IgG) - produced by plasma cells after class switching of antibodies. It can also cross the placenta and establish passive immunity.
Immunoglobulin D (IgD) - mainly found bound to B cells.
Immunoglobulin E (IgE) - activates mast cells and basophils leading to the release and formation of inflammatory mediators such as histamine, carboxypeptidase A3, tryptase, eosinophil chemotactic factor, prostaglandin D2, Leukotrienes, TNFa, and platelet-activating factor (PAF). IgE is involved in allergic reactions and immune response to helminths and protozoan pathogens.
Complement And Lectin Pathways
The innate immune system can be activated via 3 routs of the complement pathways:
Lectins and LPS pathway - trigger C4a, C4b, and C2a, C2b and later C3a and C3b
Classical pathway formation of the antigen-antibody complex - trigger C3a and C3b
Alternative pathway lipid-carbohydrate complexes found in the cell wall of bacteria and fungus - trigger C3a and C3b
Once created by one of the 3 pathways, C3b binds to multiple sites on the cell surface of the pathogen.
It then binds to receptors on the surface of the macrophage or neutrophil.
C3b is best known for its opsonizing activity because when it coats the microbe, phagocytosis activity is increased.
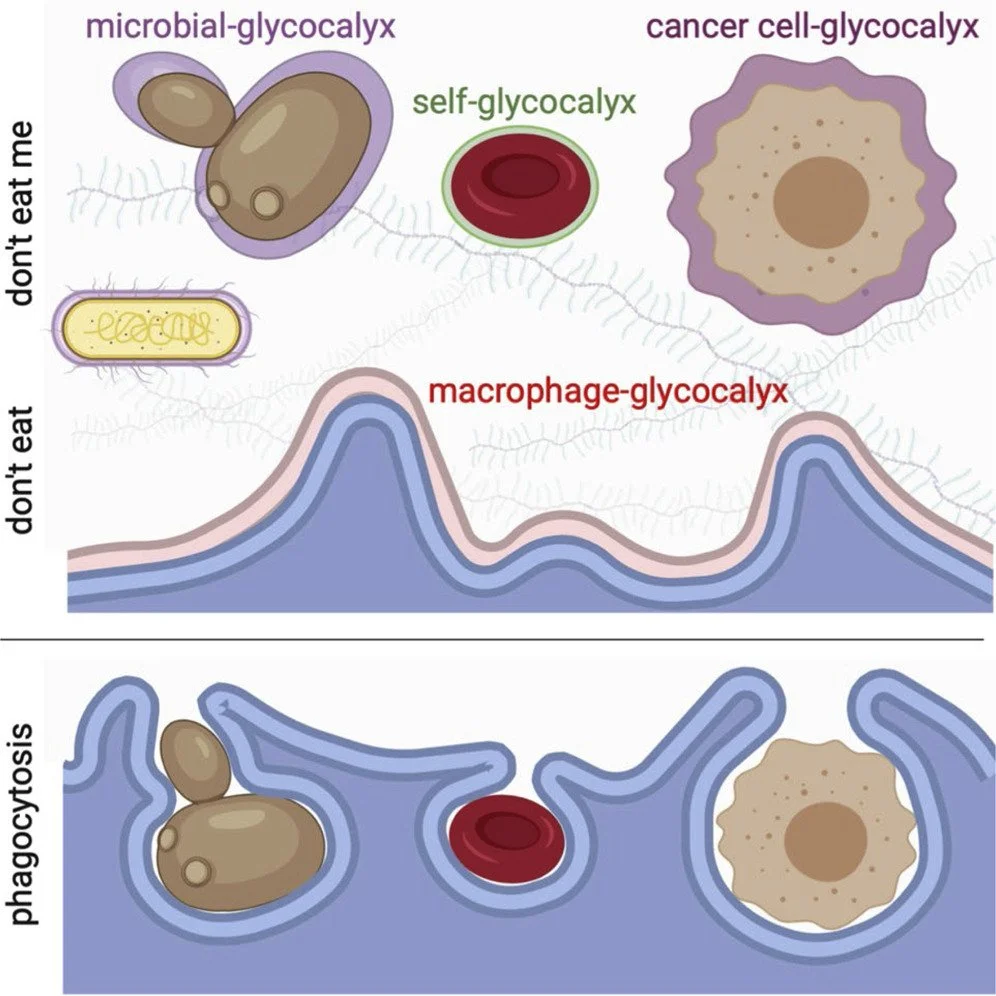
Glycosylation
The glycocalyx acts as a dysoposonin: R
on your own cells
on your cancer cells (an over abundance, that’s why phagocytes can’t find it)
on pathogens and microbes like bacteria
on your immune cells (especially phagocytes)
Having this glycocalyx layer on cancer cells and microbes makes it important to have a working innate immune system that can release proteases and cleavage enzymes (like HA-ase and MMPs) that can break down the glycocalyx.
As we discussed in the You Are Negative and That's a Positive Thing post, RBCs (zeta (ζ) potential via cholesterol sulfate), produce a dysopsonin effect.
Albumin
Albumin has a dysopsonin effect during early deposition, thus decreasing the recognition by the phagocytes, however, albumin adsorption is transient and it can be easily replaced by other proteins with higher affinities, thus having an opsonic effect. R
Antigen (Alarmin) - Antibody (Opsonin) Complexes
What’s Healthy?
When alarmins are released, they need to be cleaned up.
Your normal cleanup process is via phagocytosis.
By an alarmin (antigen) binding to an opsonin (antibody), they create a complex that is able to be recognized as an opsonin signal, that way phagocytes can come in and eat up the damage.
Molecular Mimicry or Not?
Let’s create an example here - when an alarmin is in the extracellular space (like a nucleus of a cell or a histone of DNA), your B cells pick up on that make antibodies.
When those antibodies come around they bind to anything to anything that looks like what the original alarmin was, whether it is on a healthy cell or a diseased cell.
So in this scenario, instead of binding the the nucleus or histone alarmin, it binds to a healthy cell’s nucleus or histone - this is termed molecular mimicry, yet is it really mimicking anything?
Cancer
In a cancerous cell, they have tons of dysopsonins, so antibodies are unable to tag eat me signals to it for phagocytosis.
What Do My Antibodies Mean?
As someone who had random antibodies that I couldn’t get a clean diagnosis, I had to figure this out for myself, as the classic autoimmune model just does not work.
I don’t need immunosuppressants to get better.
So what do autoimmune antibodies mean?
I will just give a few examples:
Anti Nuclear Antibodies (ANA) - the nucleus is outside the cell (generally via ETosis)
Anti Histone Antibodies (AHA) - histones of the DNA are outside the cell (generally via ETosis)
Anti DNA Antibodies (anti-dsDNA/anti-ssDNA) - DNA is outside the cell (single or double strands) (generally via ETosis)
Anti Mitochondria Antibodies (AMA) - mitochondria are outside the cell (generally via ETosis)
Anti GPCR Antibodies - receptors are floating around in the extracellular space (generally via pyroptosis or ferroptosis)
Antiphospholipid Antibodies - the lipid membrane that makes the outside of the cell is floatin the the extracellular space (generally via pyroptosis or ferroptosis)
In this sense, antibodies are your friends - they are signals to your immune system that it needs to clean up.
Do Not Eat Signalling from Viruses
Viruses have methods to evade phagocytosis by manipulating your ability to transcribe dysopsonin “don’t eat me” signals. R
Examples:
HIV-1 encodes Tat and Nef proteins, which inhibit phagocytosis of infected cells by macrophages R
Human Cytomegalovirus (HCMV) expresses the protein UL-18, which mimics the don’t-eat-me signal MHC-I α-chain to inhibit phagocytosis of the infected host cells R
Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) upregulates CD47, which inhibiting phagocytosis of the infected cells R
What makes opsonization worse?
Adjuvants - like what’s seen from vaccines, etc in Autoimmune/inflammatory syndrome induced by adjuvants (ASIA)
ETosis - Histones are among the most cardiotoxic DAMPs in sepsis R
Infections and Dysbiosis
Oxidized/Glycosylated Molecules - poor redox
PEG
Sugar - RAGE acts as a DAMP so increasing your AGEs is probably not a good idea as it continues the oxidized glycosylation cycle R
Toxins (endogenous toxins and exogenous xenobiotics)
Trauma
+/- Feedback Loops
Fixes
1/ Block Alarmins (DAMPs, PAMPs, MAMPs, etc)
First and foremost you are trying to eliminate the pathogen, toxin exposure, etc, but after you have removed yourself from exposure, you need to stop the positive feedback loop and that can be done with Suppressing/Inhibiting DAMPs (SAMPs), such as SPMs.
SPMs (Pro-Resolving Mediators)
Olive Leaf Extract (Oleuropean and Hydroxytyrosol) R
2/ Antibodies
You want to ramp up the antibodies, but remember as you do that you will experience increased phagocytosis side effects.
Mechanistic Fixes
Here are more mechanistic examples: R
HMGB1 - HMGB1 neutralizing antibodies; Heparin, heparin variants, or heparinoids
CIRP - Oligopeptide C23
Histones - Histones and their fragments; Drotrecogin alfa (activated)
ATP, ADP, AMP - Alkaline phosphatase (ALP)
DNA - DNAses
mitochondrial DAMPs - Quinone-based antioxidants (like hydroxycholroquinone); Long omega-3 polyunsaturated fatty acids (like SPMs); Deformylase
LPS - ALP; Synthetic anti-lipopolysaccharide peptides (SALPs)
Flagellin - Anti-flagellin sub-type monoclonal antibody
TLR4 -Alkaline phosphatase (ALP); Eritoran; Resatorvid (TAK242); Small agonists/antagonists (e.g., MPLA)
NLRP3 - MC950; Melatonin
P2 - AZ 10606120; Suramin
RAGE - Anti-RAGE antibodies; siRNAs; Soluble RAGE (sRAGE)
TREM1 - Soluble TREM-1 (Nangibotide)